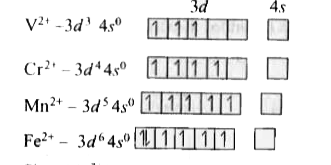
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
THE D- AND F- BLOCK ELEMENTS
DISHA PUBLICATION|Exercise EXERCISE - 1 CONCEPT BUILDER (TOPIC 3: LANTHANOIDS AND ACTINOIDS)|20 VideosSURFACE CHEMISTRY
DISHA PUBLICATION|Exercise Exercise-2 :Concept Applicator|30 VideosThe p-Block Elements (Group 13 and Group 14)
DISHA PUBLICATION|Exercise Exercise-2: Concept Applicator|30 Videos
Similar Questions
Explore conceptually related problems
DISHA PUBLICATION-THE D- AND F- BLOCK ELEMENTS -EXERCISE - 2 CONCEPT APPLICATOR
- The correct order of magentic moments (spin only values is B.M ) among...
Text Solution
|
- The basic character of the transition metal monoxides follows the orde...
Text Solution
|
- Four successive members of the first row transition elements are liste...
Text Solution
|
- Which of the following does not represent the correct order of the pro...
Text Solution
|
- Zn and Hg do not show variable valency like d-block elements because-
Text Solution
|
- Which of the following statement is incorrect
Text Solution
|
- A compound of iron exists as a dimer in vapour state. It is hygroscopi...
Text Solution
|
- Which of the following statements is wrong ?
Text Solution
|
- A metal M and its compounds can gives the following observable changes...
Text Solution
|
- Which tow sets of reactants best represents the amphoteric character o...
Text Solution
|
- Copper is very slowly oxidised on the surface in moist air, givinga gr...
Text Solution
|
- A red solid is insoluble in water. However, it becomes soluble if some...
Text Solution
|
- Which of the following reactions represents " developing"in photograph...
Text Solution
|
- In the extraction of silver from argentite ore the ore is treated witl...
Text Solution
|
- The dichromate ion is in cquilibrium with chromate ion in aqucous Solu...
Text Solution
|
- A blue colouration is not obtained when
Text Solution
|
- When potassium ferrocyanide crystals are heated with conc. sulphuric a...
Text Solution
|
- Precipitate of AgCl is soluble in liquid NH(3) the compound forms
Text Solution
|
- Which of the following products are obtained when Na2CO3 is added to a...
Text Solution
|
- Which of the following statement is not correct?
Text Solution
|