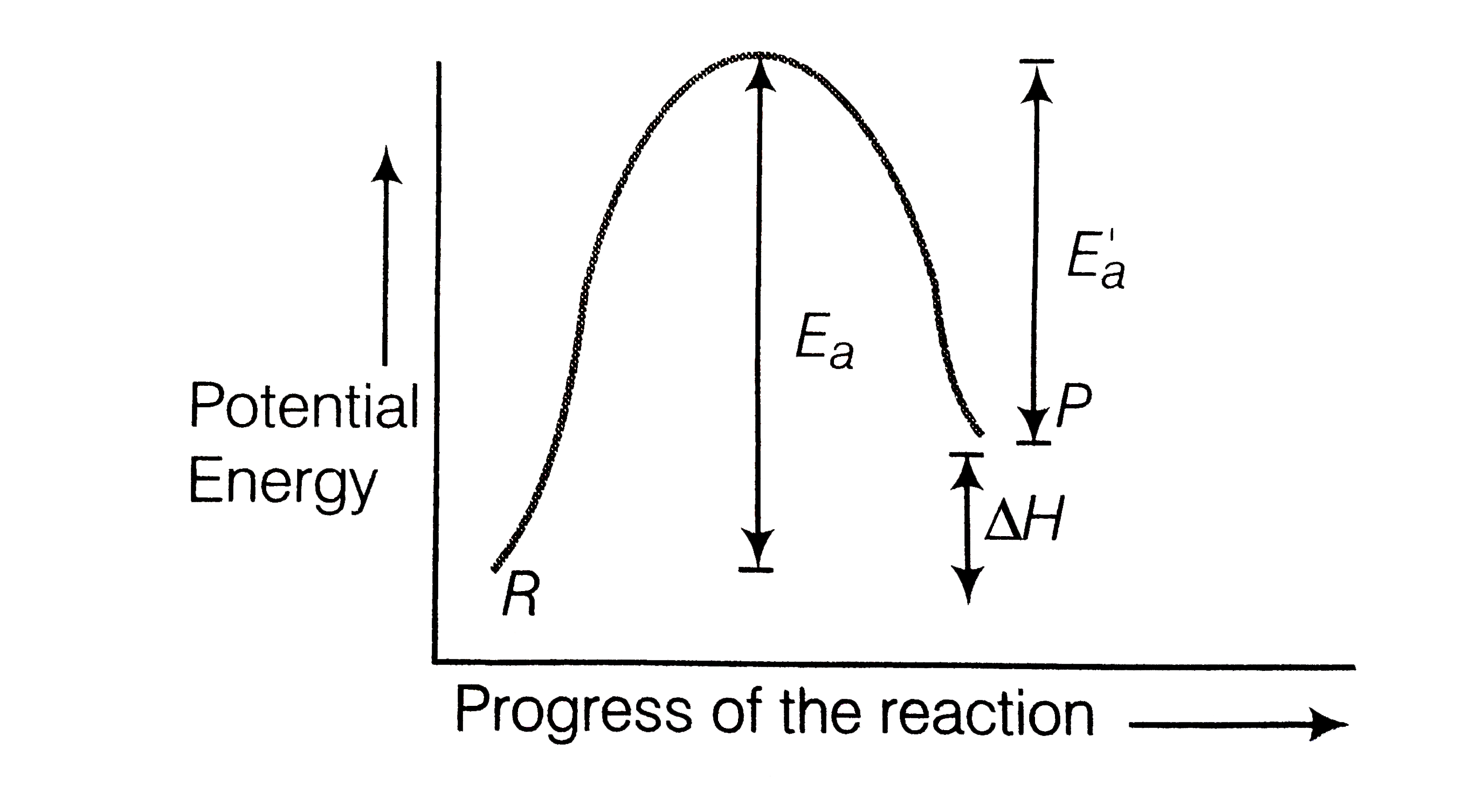
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- For an endothermic reaction energy of activation is E(a) and enthlpy o...
Text Solution
|
- A colliison between reactant molecules must occur with a certain minim...
Text Solution
|
- For an endothermic reaction, where Delta H represents the enthalpy of ...
Text Solution
|
- For an endothermic reaction energy of activation is E(a) and enthlpy o...
Text Solution
|
- For an endothermic reaction , energy of activation is E(a) and enthalp...
Text Solution
|
- An endothermic reaction have an activation energy of E(a)kJ.mol^(-1) ...
Text Solution
|
- Following are the values of E(a) and Delta H for three reactions carri...
Text Solution
|
- For an endothermic reaction, energy of activation is Ea and enthalpy o...
Text Solution
|
- For an endothermic reaction, where Delta H represents the enthalpy of ...
Text Solution
|