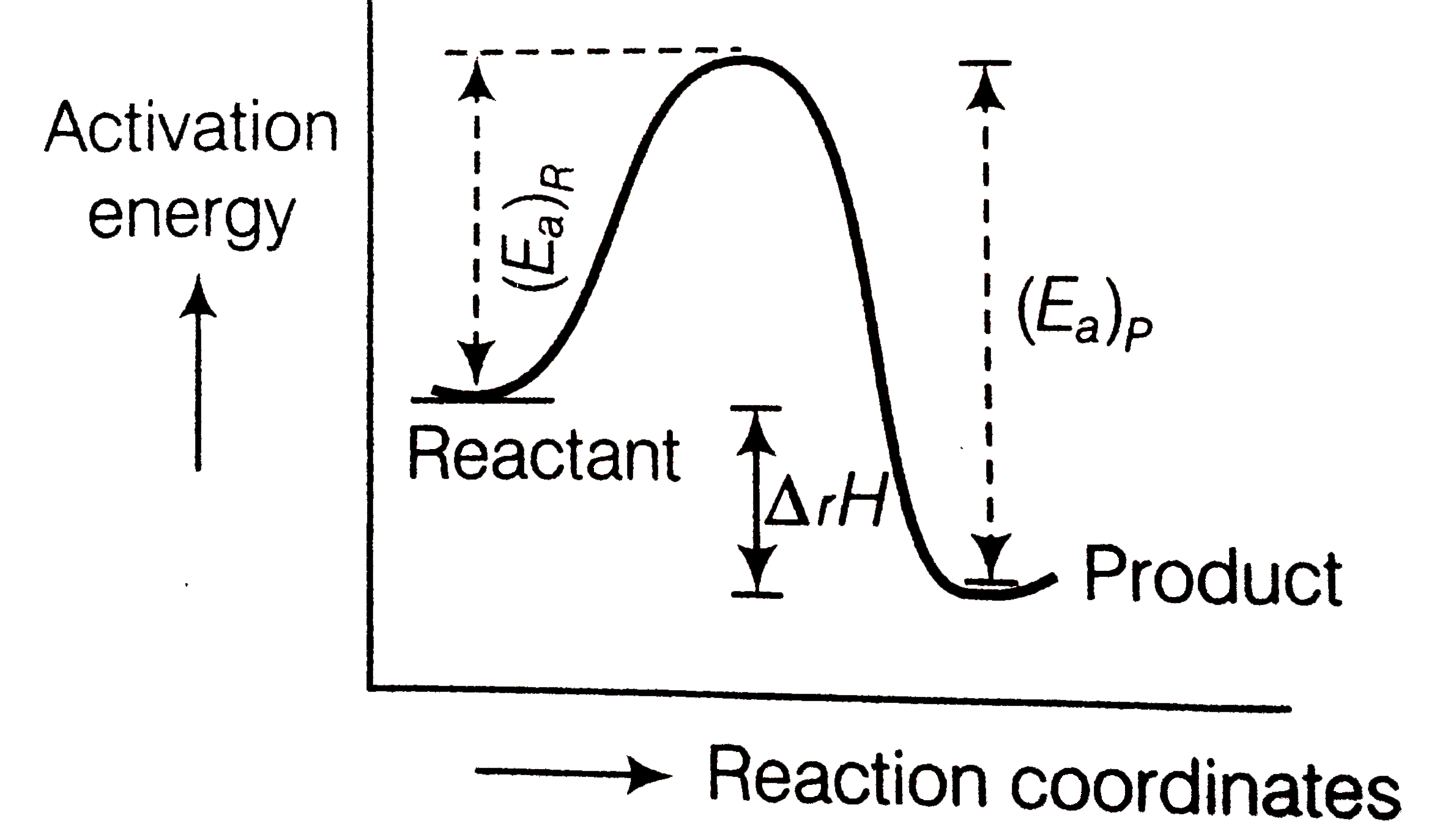A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- For an exothermic reaqction, the energy of activation of the reactants...
Text Solution
|
- Statement: A catalyst always lower the energy of ectivation. Explana...
Text Solution
|
- Assertion: An exothermic reaction in principle cannot have zero active...
Text Solution
|
- For an exothermic reaqction, the energy of activation of the reactants...
Text Solution
|
- In exothermic reactions, the energy of products is more than the energ...
Text Solution
|
- Energy of activation of a reactant is reduced by
Text Solution
|
- सक्रियण ऊर्जा का मान ऊप्माक्षेपी अभिक्रियाओं में अधिक होता है अथवा ऊष्...
Text Solution
|
- Energy of activation of an exothermic reaction reaction is .
Text Solution
|
- Energy of activation of an exothermic reaction reaction is .
Text Solution
|