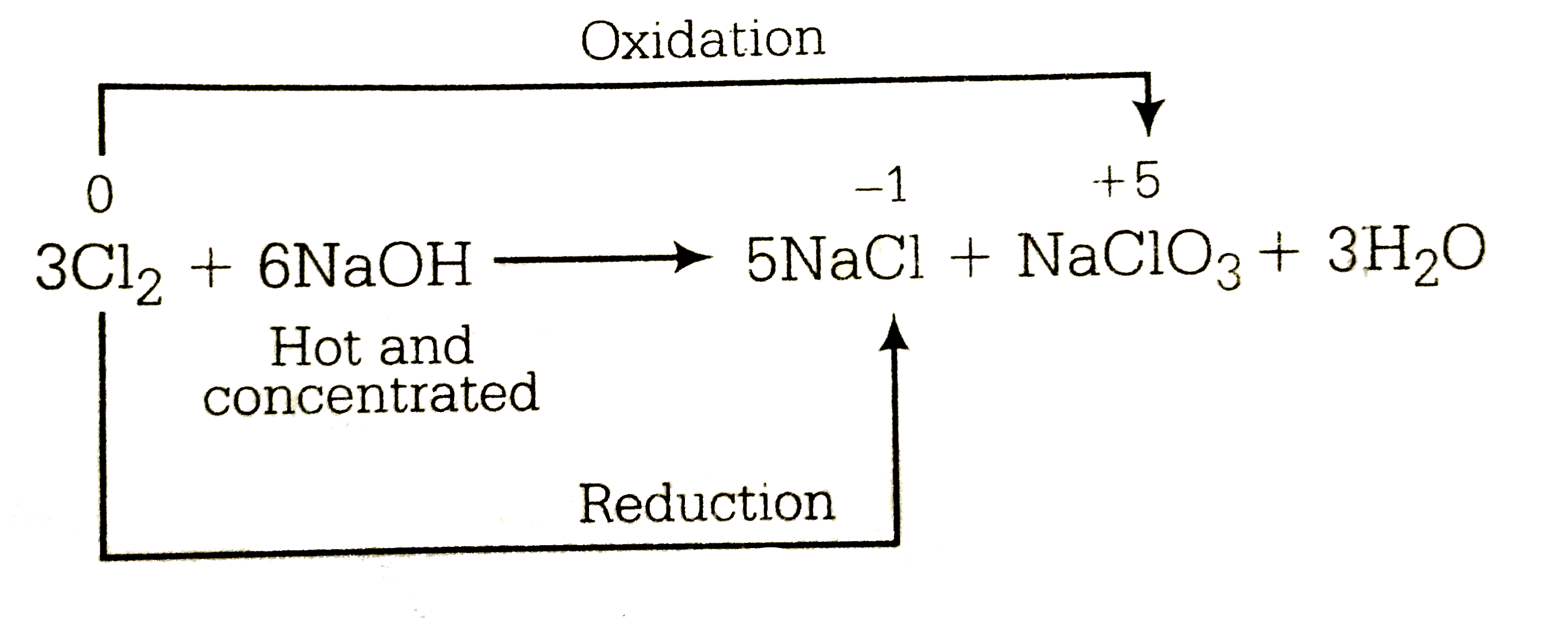A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- When Cl(2) gas reacts with hot and concentrated sodium hydroxide solut...
Text Solution
|
- When Cl(2) gas reacts with hot and concentrated sodium hydroxide solut...
Text Solution
|
- When CI(2) gas reacts with hot and concentrated sodium hydroxide solut...
Text Solution
|
- When CI(2) gas reacts wioth hot and concentrated solution of sodium hy...
Text Solution
|
- क्लोरीन की ठण्डे व तनु एवं गर्म व सान्द्र सोडियम हाइड्रॉक्साइड विलयन स...
Text Solution
|
- जब CI(2) और गर्म तथा सान्द्र सोडियम हाइड्रोऑक्साइड विलयन के साथ अभिक्र...
Text Solution
|
- When Cl(2) gas reacts with hot and concentrated sodium hydroxide solut...
Text Solution
|
- When Cl(2) gas reacts with hot and concentrated sodium hydroxide solut...
Text Solution
|
- When CI(2) gas reacts with hot and concentrated sodium hydroxide solut...
Text Solution
|