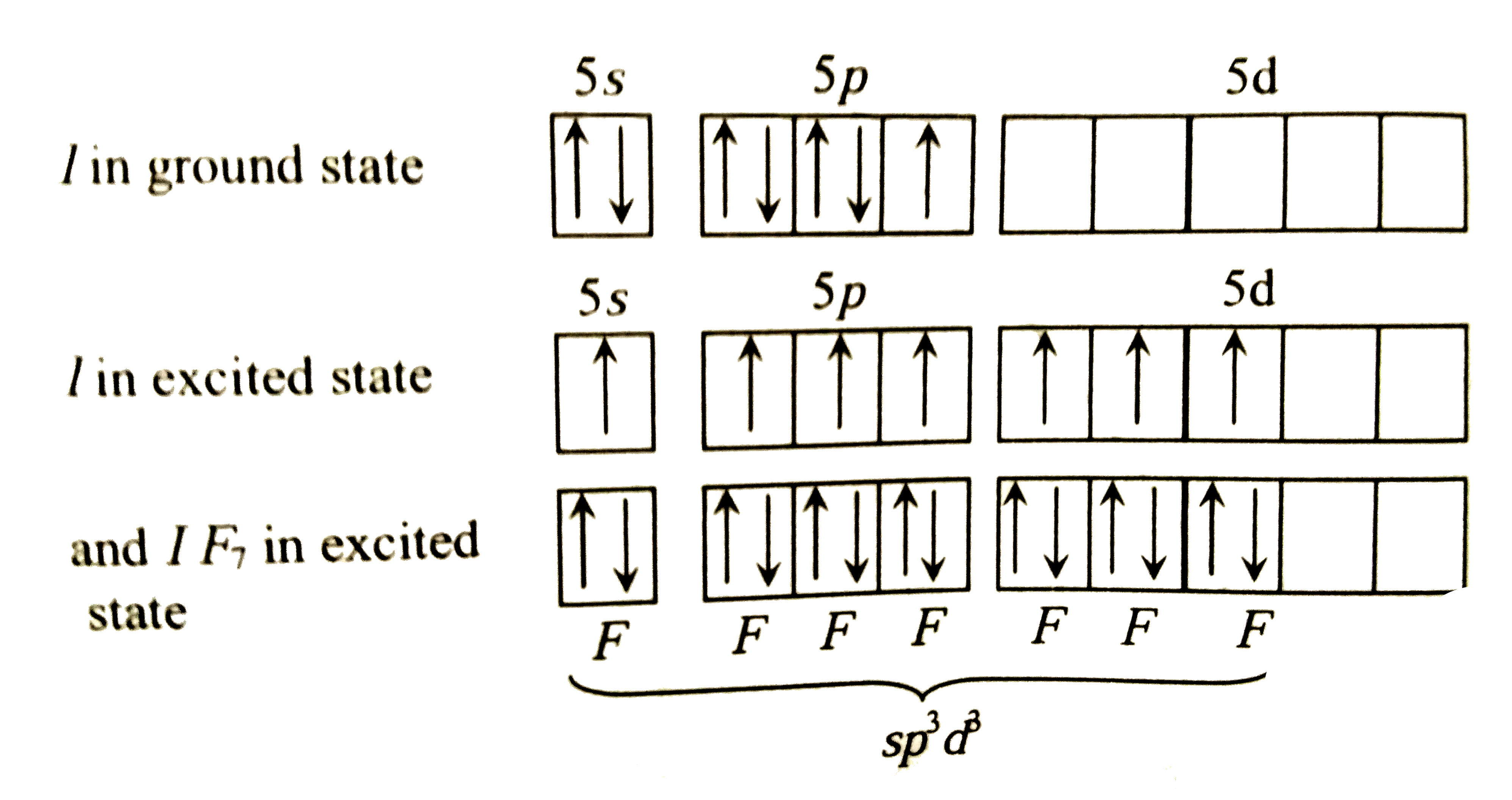A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- The structure of IF(7) is
Text Solution
|
- The structure of IF(7) is
Text Solution
|
- The structure of IF(7) is
Text Solution
|
- Draw structure and give hybridisation of IF(7).
Text Solution
|
- The structure of IF(7) is
Text Solution
|
- IF(7) की संरचना है :
Text Solution
|
- Explain the structures of IF(7)
Text Solution
|
- The structure of IF(7) is
Text Solution
|
- The structure of IF(7) is-
Text Solution
|