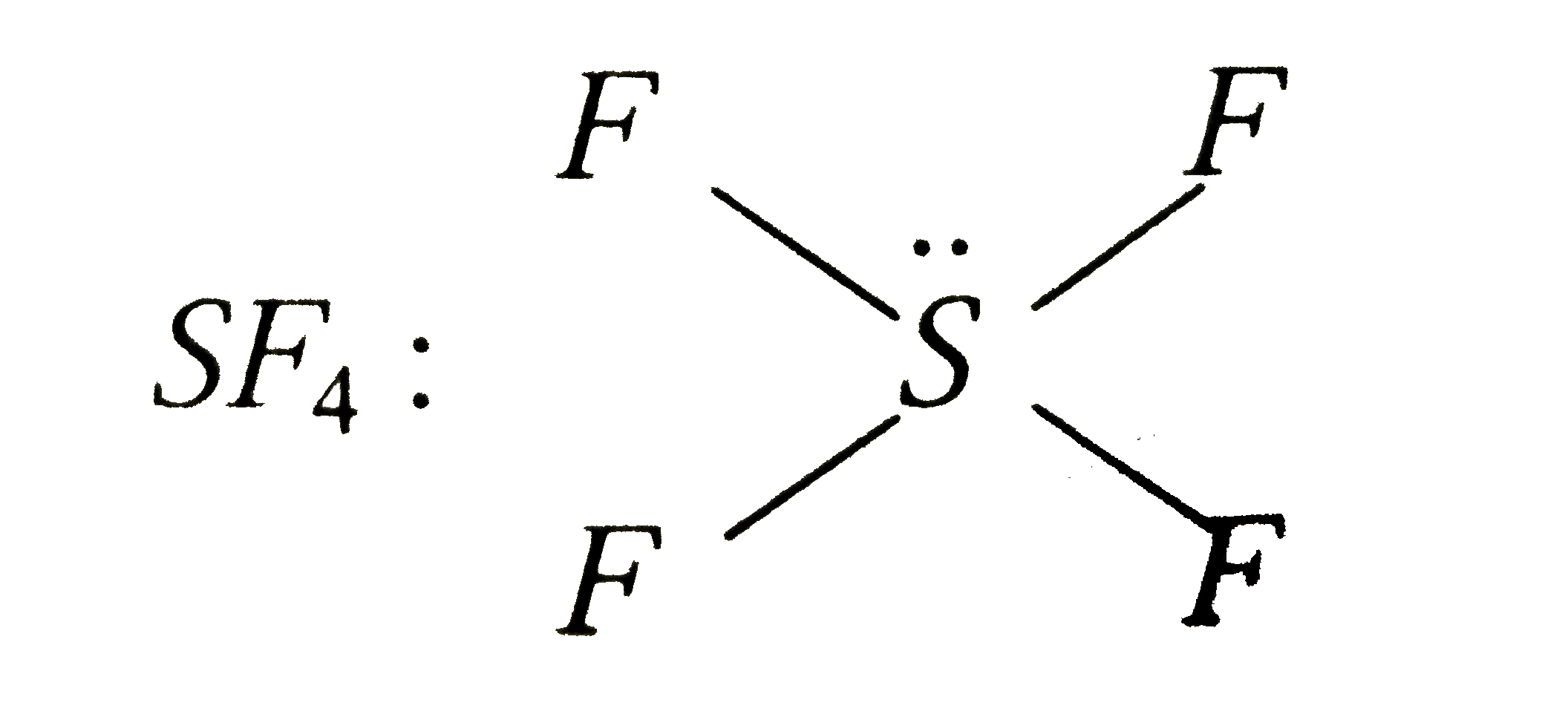
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- SF(2),SF(4) and SF(6) have the hybridisation at sulphur atom respectiv...
Text Solution
|
- SF(2),SF(4) and SF(6) have the hybridisation at sulphur atom respectiv...
Text Solution
|
- Assertion:- SF(6) cannot be hydrolysed but SF(4) can be. Reason:- Six ...
Text Solution
|
- Assertion :- SF(6) cannot be hydrolysed SP(4) can be Reason :- Six F a...
Text Solution
|
- The hybridisation of sulphur in SF(4) is
Text Solution
|
- The hybridisation of sulphur in SF(6) is
Text Solution
|
- Statement 1: SF(6) molecular has octahedral geometry Statement 2: Su...
Text Solution
|
- Atomic orbitals involved in hybridisation of SF(6) molecule
Text Solution
|
- SF(2), SF(4) and SF(6) have the hybridisations at sulphur atom respect...
Text Solution
|