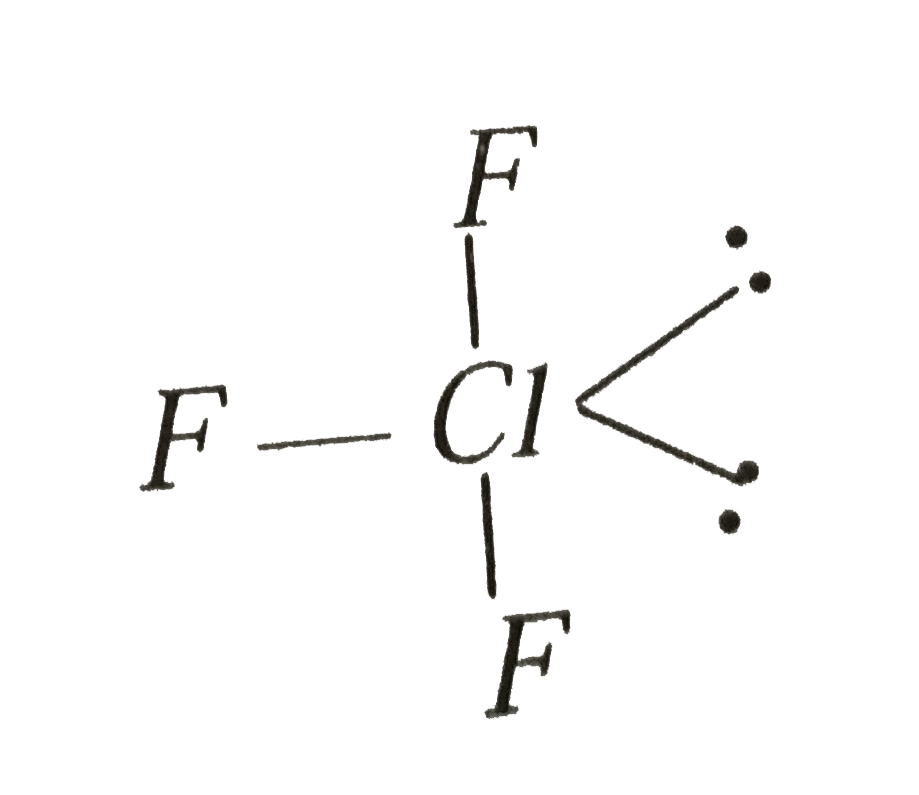A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- According to VSEPR theory, the shape of the water molecule is
Text Solution
|
- The shape of ClO(3)^(-) ion according to VSEPR theory is:
Text Solution
|
- According to VSEPR theory, the shape of the water molecule is
Text Solution
|
- According to VSEPR theory, the shape of OF(2) molecule is .
Text Solution
|
- The shape of XeF4 molecule according to VSEPR theory is
Text Solution
|
- Predict the shape of ClF(3) according to VSEPR theory.
Text Solution
|
- VSEPR सिद्धान्त के अनुसार, जल के अणु की आकृति होती है
Text Solution
|
- VSEPR theory is used to predict the shape of covalent molecules . a) S...
Text Solution
|
- VSEPR theory is used to predict the shape of covalent molecules . b) ...
Text Solution
|