A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
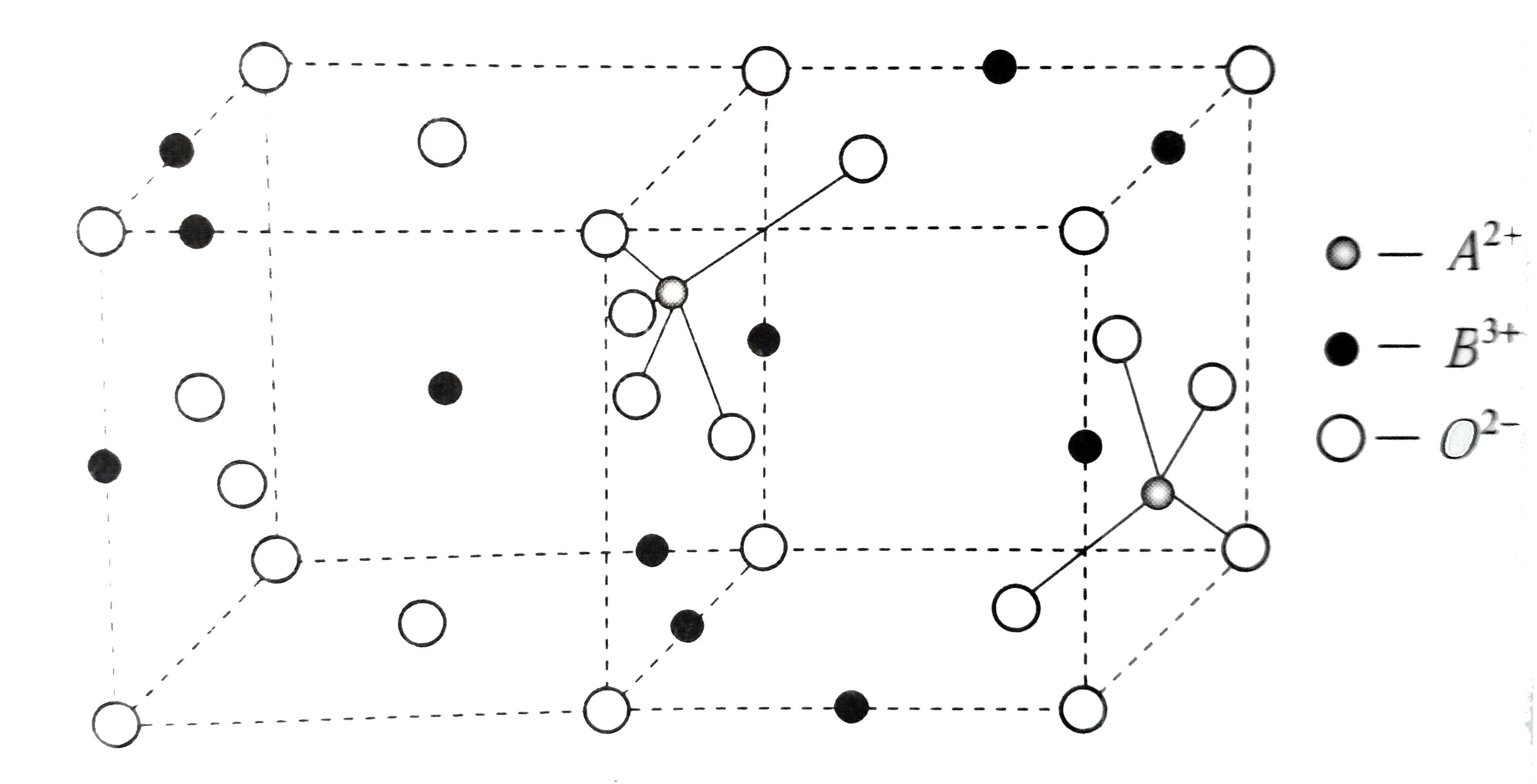
- A class of oxide is a spinal , it consists of two types of metal ions ...
Text Solution
|
- A spinel is an important class of oxide consisting two types of ...
Text Solution
|
- A spinel is an important class of oxide consisting two types of ...
Text Solution
|
- A class of oxide is a spinal , it consists of two types of metal ions ...
Text Solution
|
- A class of oxide is a spinal , it consists of two types of metal ions ...
Text Solution
|
- A class of oxide is a spinal , it consists of two types of metal ions ...
Text Solution
|
- A class of oxide is a spinal , it consists of two types of metal ions ...
Text Solution
|
- Spinel is a important class of oxides consisting of two types of metal...
Text Solution
|
- Spinel is a important class of oxides consisting of two types of metal...
Text Solution
|