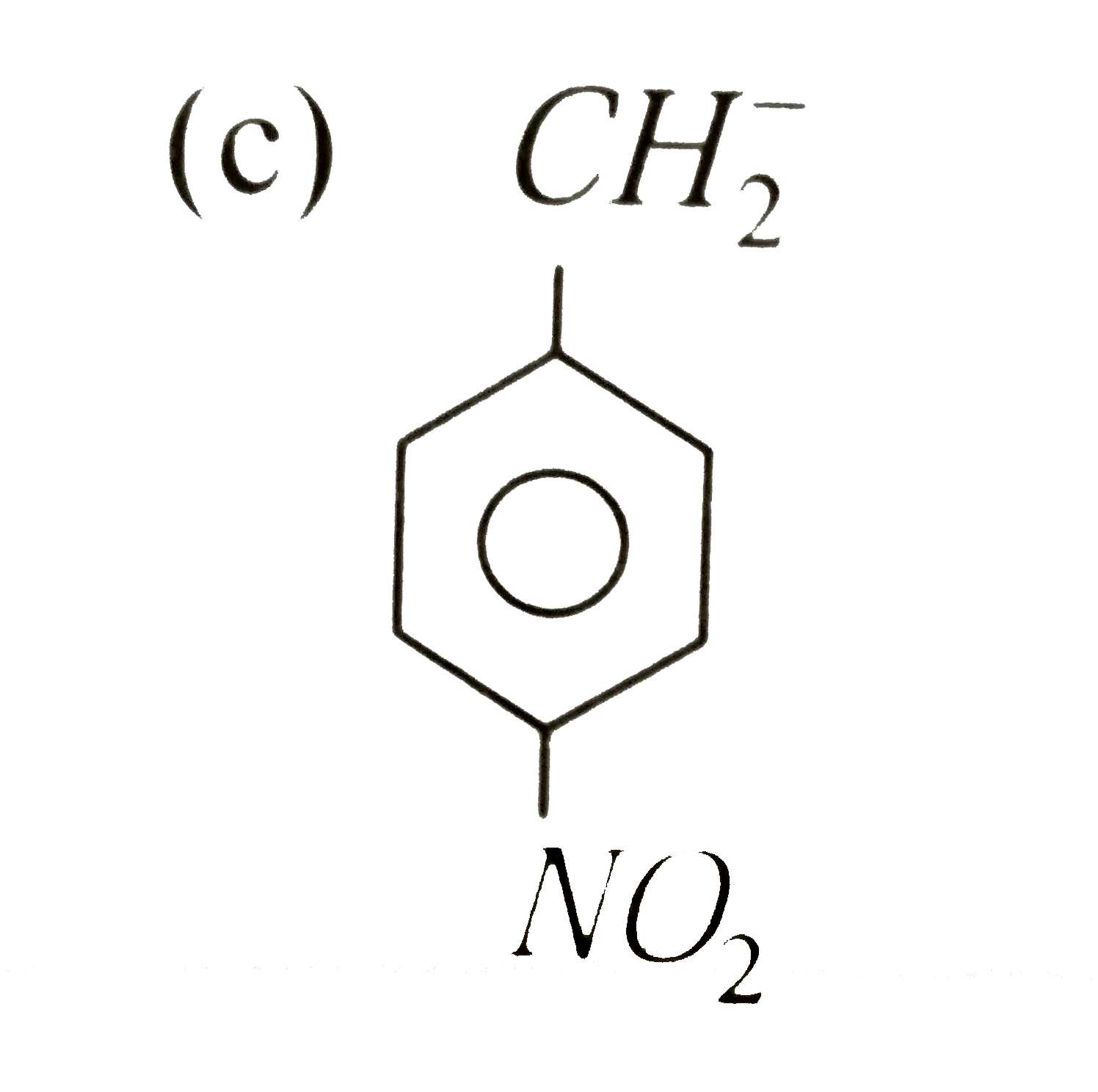
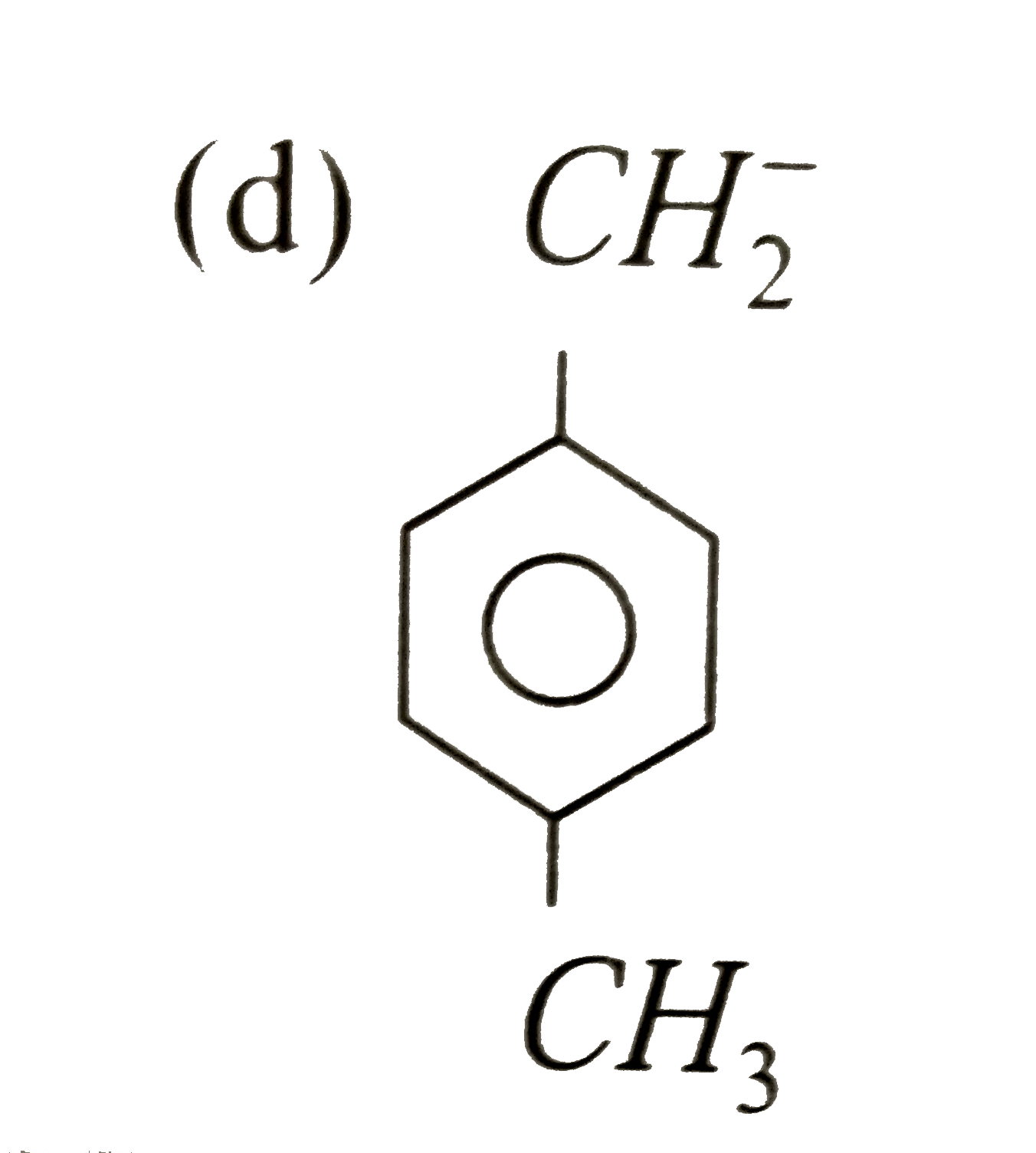A
B
C
D
Text Solution
AI Generated Solution
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- Most stable carbanion is
Text Solution
|
- Most stable carbanion is :-
Text Solution
|
- Which of the folowing carbanions is most stable?
Text Solution
|
- Which of the following carbanion is most stable ?
Text Solution
|
- The most stable carbanion is :
Text Solution
|
- Most stable carbanion is
Text Solution
|
- The most stable carbanion is
Text Solution
|
- Which one of the carbanions is the most stable-
Text Solution
|
- The most stable carbanion is-
Text Solution
|