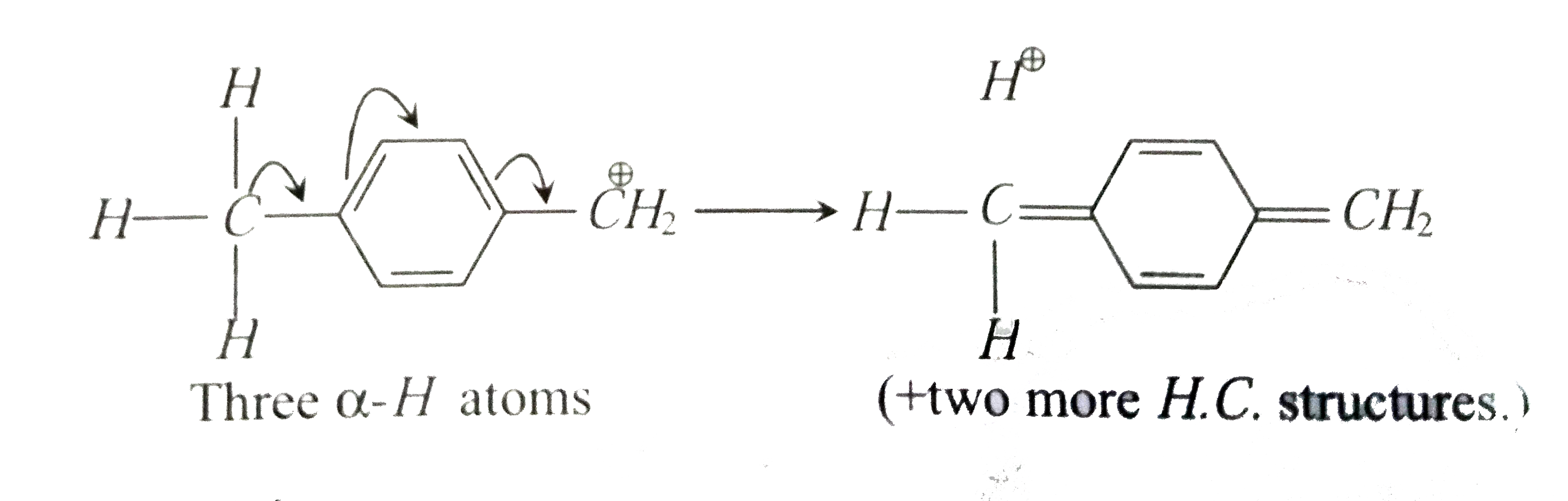A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- p-methyl benzyl carbocation (I) is more stable than benzyl carbocation...
Text Solution
|
- APPLICATIONS OF HYPERCONJUGATION-STABILITY OF BENZYL CARBOCATION
Text Solution
|
- p-methyl benzyl carbocation (I) is more stable than benzyl carbocation...
Text Solution
|
- Assertion (A): The major products formed by heating with HI are: Reaso...
Text Solution
|
- Statement-I: Alkenes are more reactive than alkynes towards HX. Statem...
Text Solution
|
- Benzyl carbocation is more stable than ethyl carbocation. Justify.
Text Solution
|
- Out of benzyl and ethyl carbocation which is more stable?
Text Solution
|
- Statement-I: The major products formed by heating with HI are St...
Text Solution
|
- Arrange the following carbocations in order of stability {:("benzyl ...
Text Solution
|