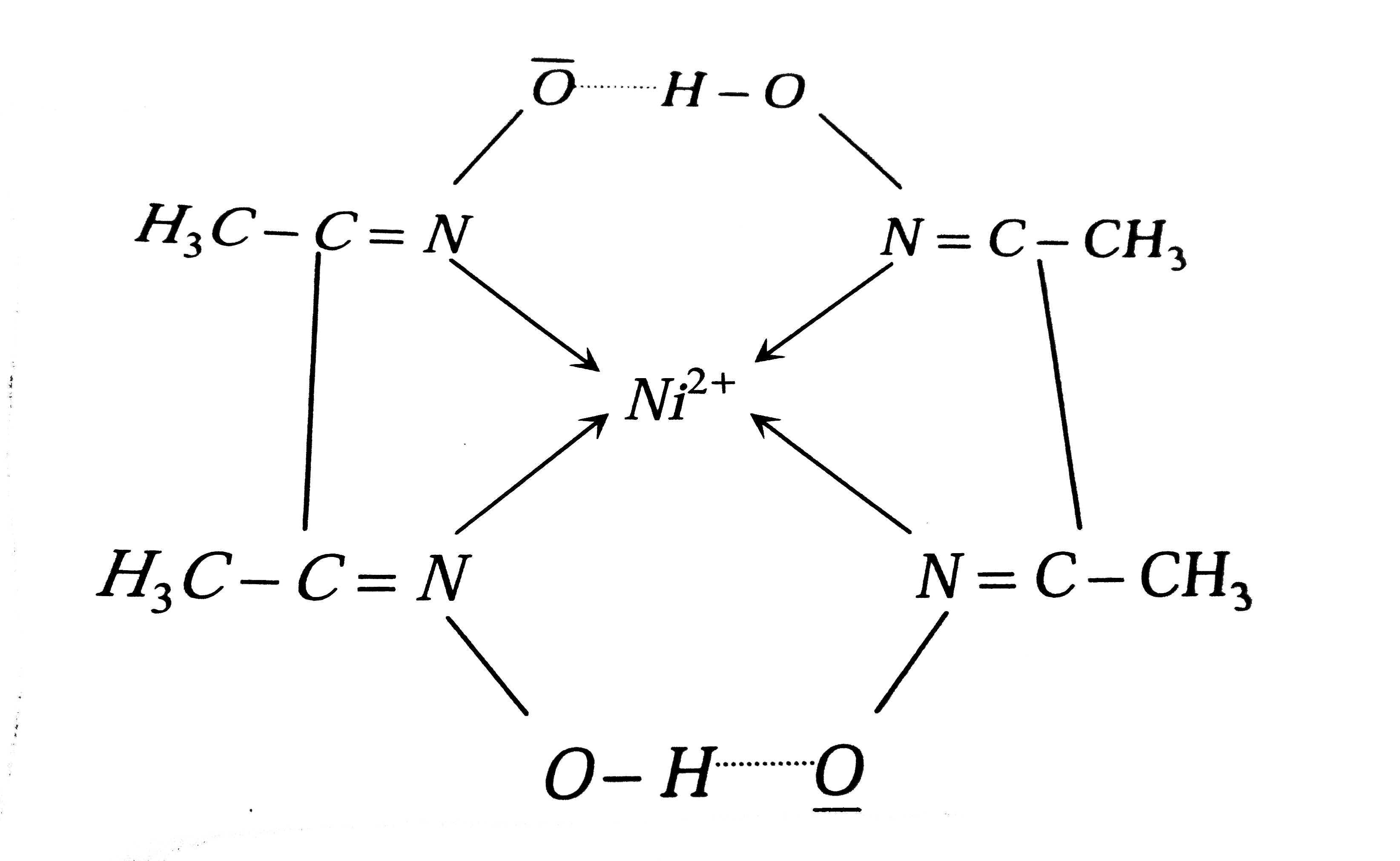
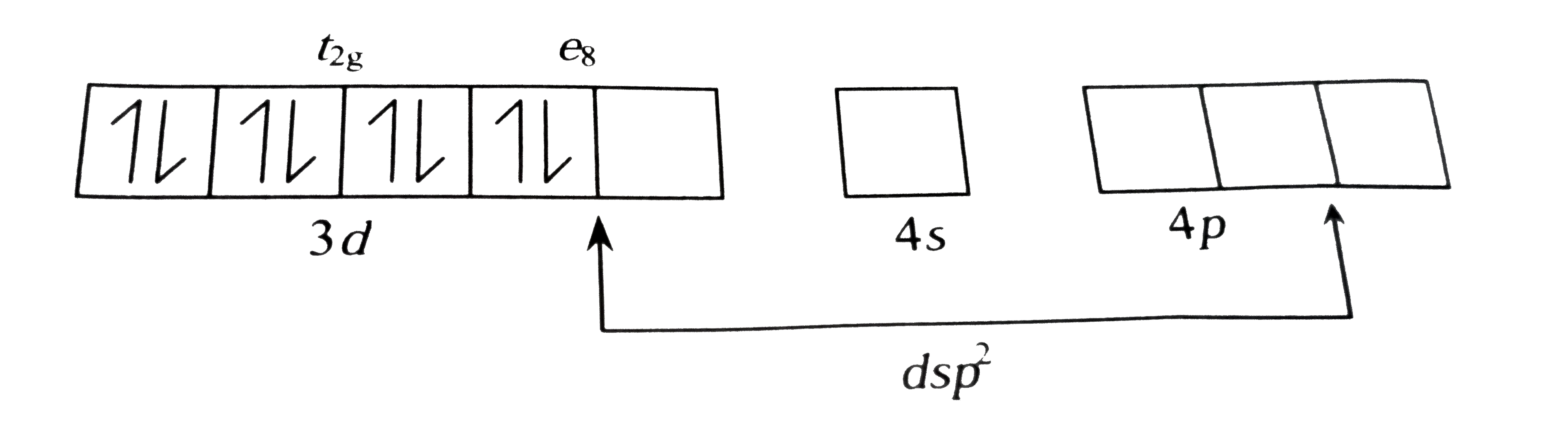
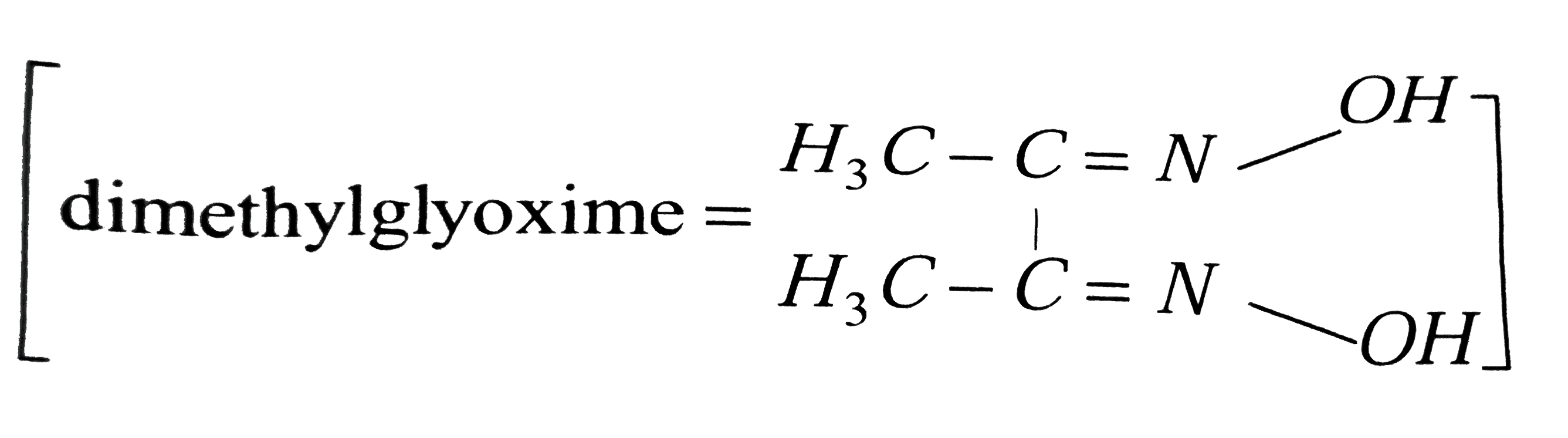A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- Red precipitae is obtained when ethanol solution of dimethylglyoxime i...
Text Solution
|
- Red precipitae is obtained when ethanol solution of dimethylglyoxime i...
Text Solution
|
- Which one of the following gives a red precipitate with ammoniacal sol...
Text Solution
|
- When dimethylglyoxime is added to Ni^(2+), which of the following stat...
Text Solution
|
- Red precipitate is obtained when enthanol solution of dimethylglyoxime...
Text Solution
|
- जब डाइ मीथायालाग्लायोक्साइम के एल्कोहोलिक को अमोनियाकल Ni(II) में से म...
Text Solution
|
- जब डाईमेथिलग्लाइऑक्जीम के एल्कोहलिक विलयन को अमोनिकल Ni(II) में मिलाया...
Text Solution
|
- Which of the following gives a red precipitate with an ammoniacal solu...
Text Solution
|
- Addition of ethanol solution of dimethylglyoxyme to ammonium-containin...
Text Solution
|