A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
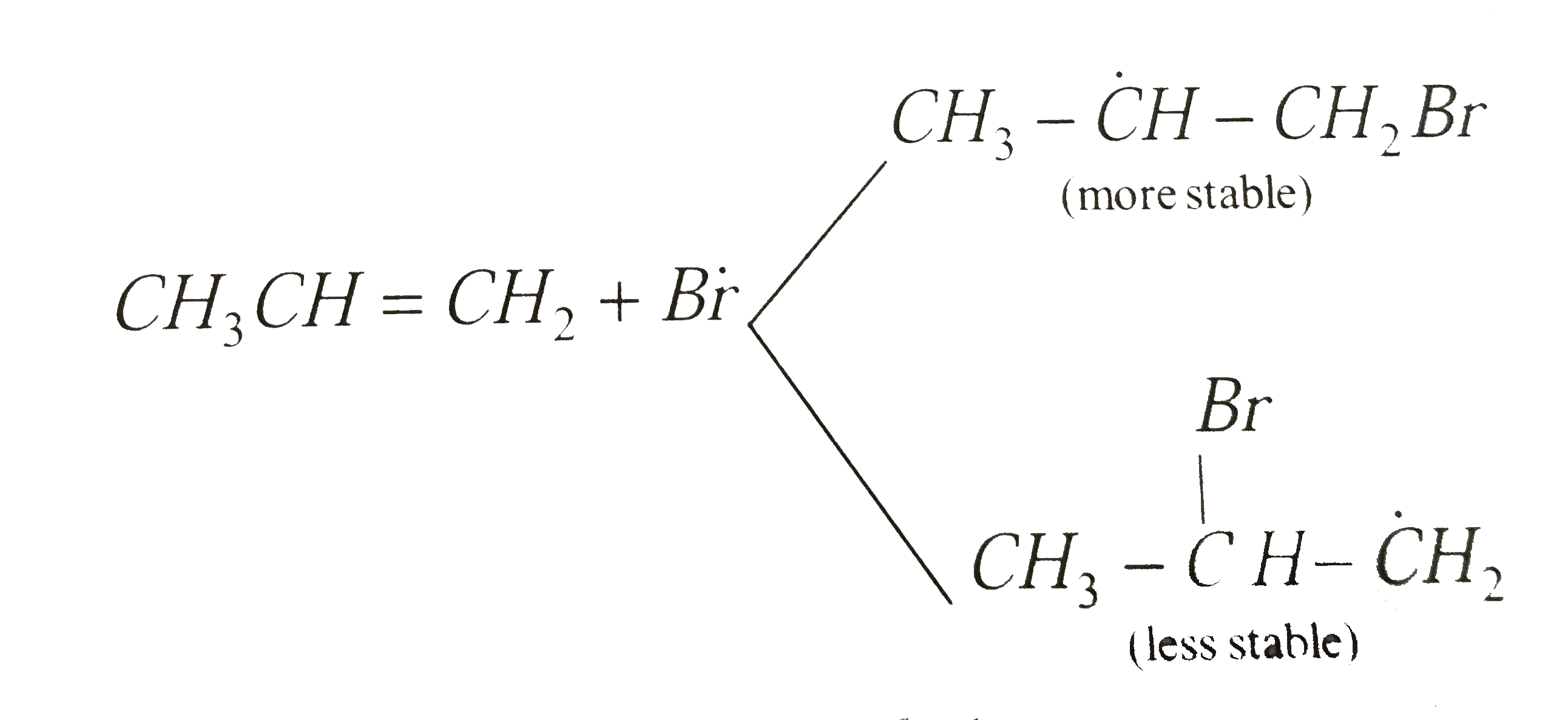
- Reaction of HBr with propene in the presence of peroxide gives :-
Text Solution
|
- The reaction of propene with HBr in presence of peroxide proceeds thro...
Text Solution
|
- 3-phenyl propene on reaction with HBr in the presence of peroxide, the...
Text Solution
|
- Reaction of HBr with propene in the presence of peroxide gives
Text Solution
|
- Reaction of HBr with propene in the presence of peroxide gives :-
Text Solution
|
- The reaction of propene with HBr in presence of peroxide proceeds thro...
Text Solution
|
- The reaction of HBr with 1-propene in the presence of peroxides will p...
Text Solution
|
- Propene reacts with HBr in the presence of a peroxide to form :
Text Solution
|
- The addition of HBr to propene in the presence of peroxides follows .....
Text Solution
|