A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- Statement-I : Nitration of toluene is easier than benzene Because ...
Text Solution
|
- S-I: Toluene on Friedel craft's methylation gives o- and p-xylene S-II...
Text Solution
|
- Statement-I : Propene is more reactive than ethnc with HCl . Because S...
Text Solution
|
- Statement-I : Nitration of toluene is easier than benzene Because ...
Text Solution
|
- Statement-I : The rate of nitration of benzene is less than hexa deute...
Text Solution
|
- Statement-I : Nitration of toluene is easier than benzene Because ...
Text Solution
|
- The vapour pressure of pure benzene and toluene are 160 and 60 torr re...
Text Solution
|
- Why phenol undergoes electrophilic substitution more easily than benze...
Text Solution
|
- Toluene undergoes nitration easily than Benzene why?
Text Solution
|
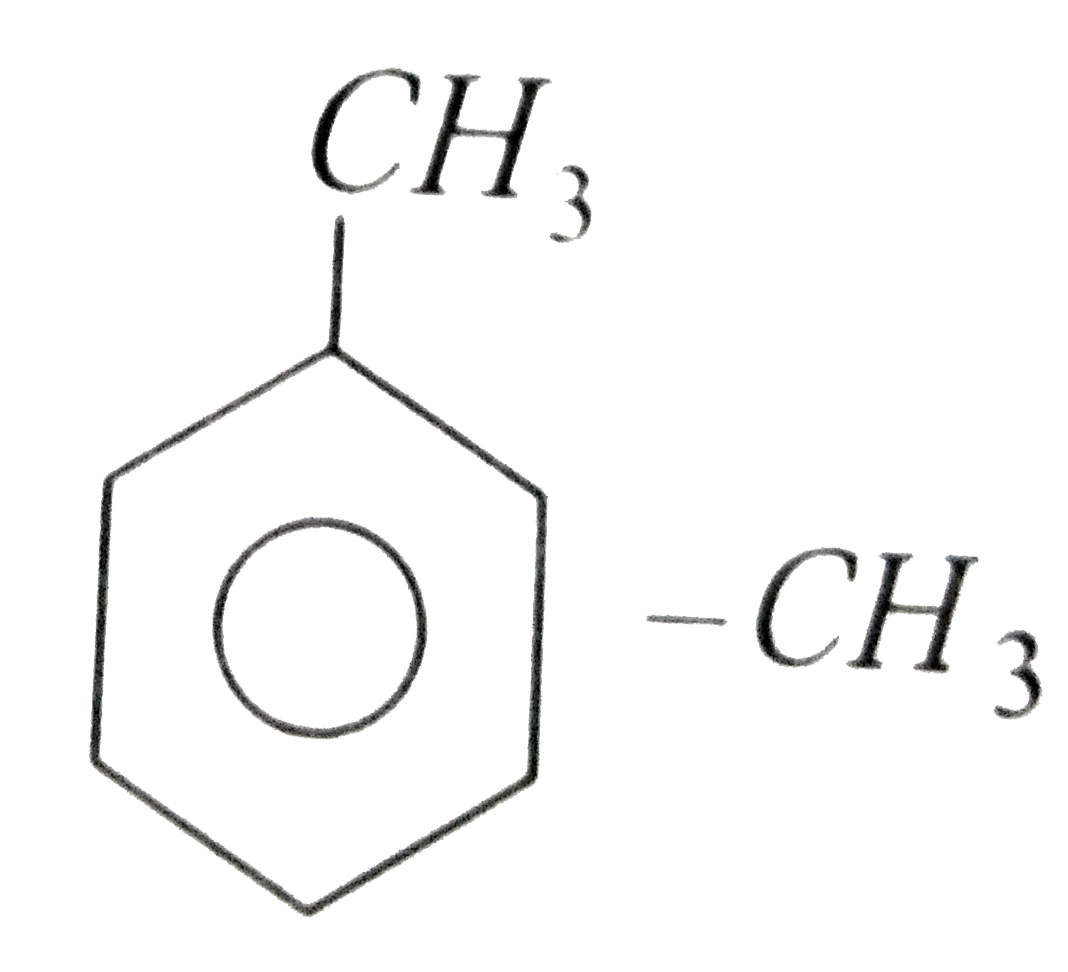 group is electron Pushing . It is an activating group. By its+ I effect, `-CH_3` group activates the benzene ring at ortho and para position relative to its. Due to increased activity towards electrophilic substitution toluene can be more easily nitrated than benzene.
group is electron Pushing . It is an activating group. By its+ I effect, `-CH_3` group activates the benzene ring at ortho and para position relative to its. Due to increased activity towards electrophilic substitution toluene can be more easily nitrated than benzene.