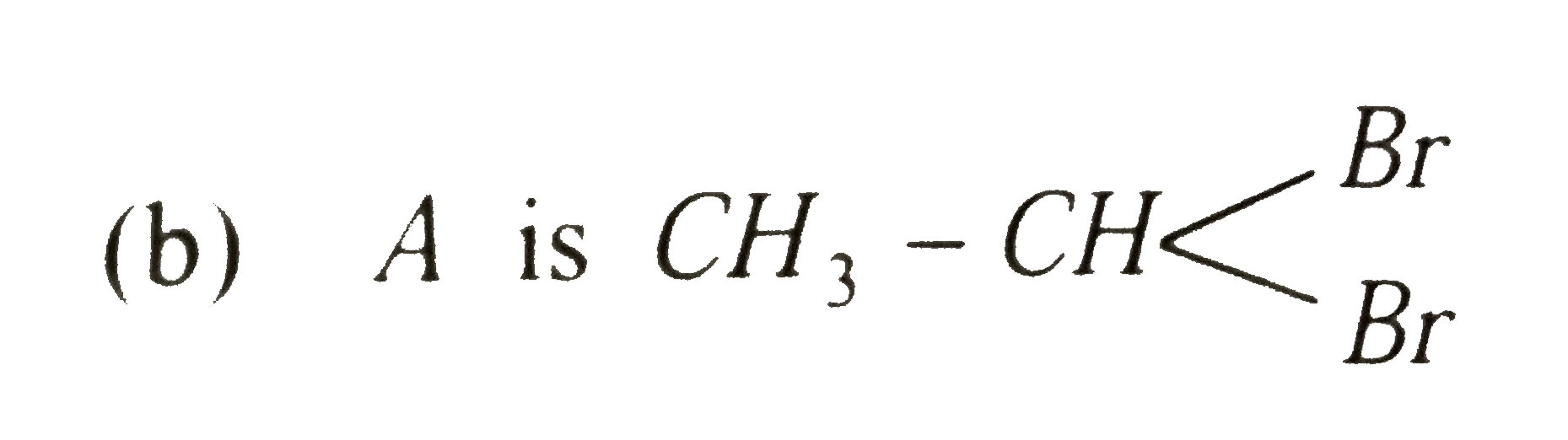A
B
C
D
Text Solution
AI Generated Solution
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- When CH2=CH-Br is reacted with HBr then the product formed is A and wh...
Text Solution
|
- The reaction of one equivalent of HBr with CH2=CH-CH2-C-=CH gives :
Text Solution
|
- Ph-CH=CH2+HBr "Peroxide"to Major product is :
Text Solution
|
- When CH2=CH-Br is reacted with HBr then the product formed is A and wh...
Text Solution
|
- CH3 -CH=CH2 +HBr to …….निर्मित उत्पाद होगा
Text Solution
|
- Which of the following compounds is formed on addition of HBr to CH2 =...
Text Solution
|
- CH2 = CH- CH= CH2 overset(HBr( 1 " mole"))to major product
Text Solution
|
- The product of the reaction CH3-CH=CH2 +HBr rarr (X) is :
Text Solution
|
- The product of the reaction CH3-CH=CH2 +HBr rarr (X) is :
Text Solution
|