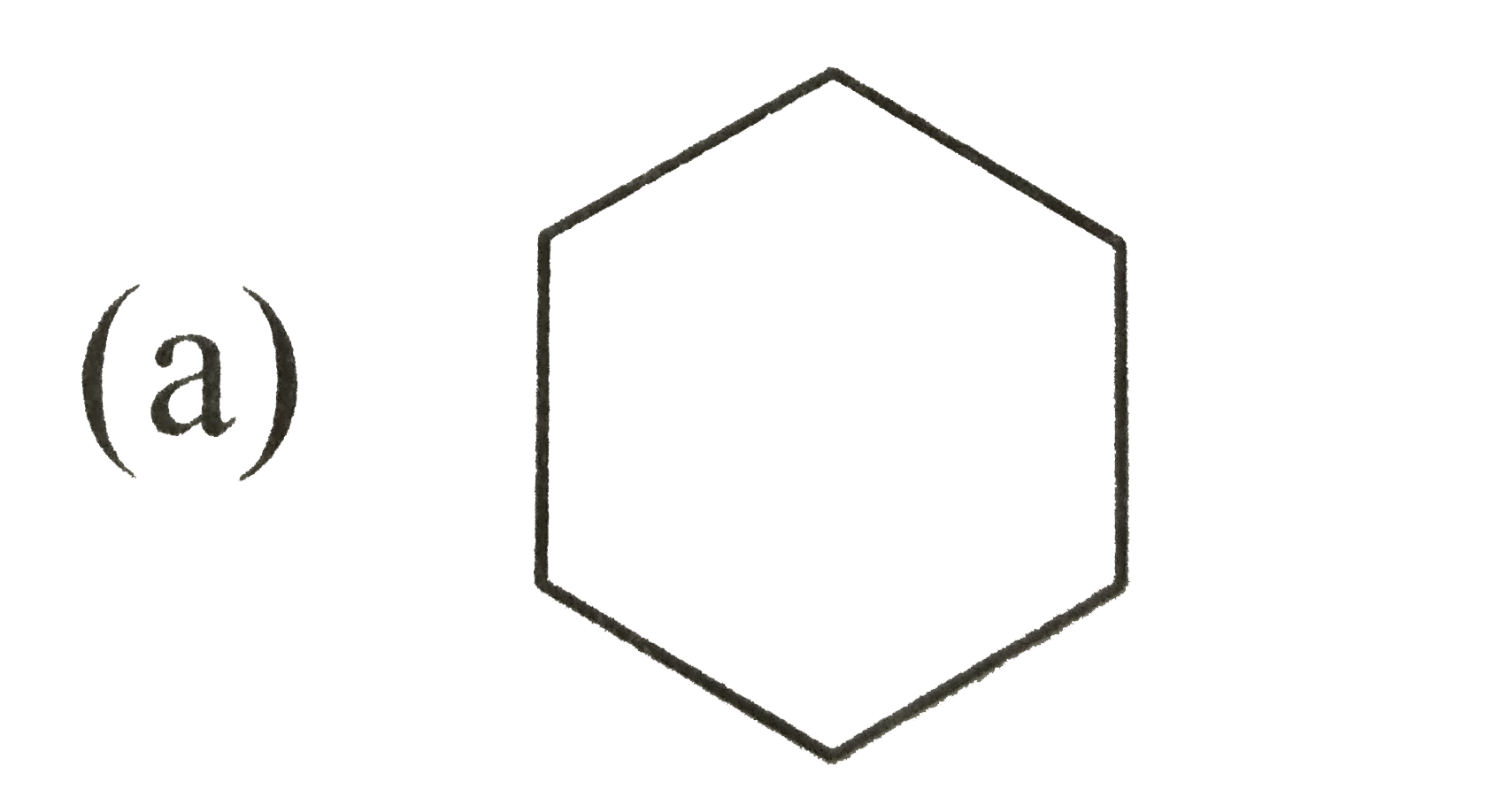
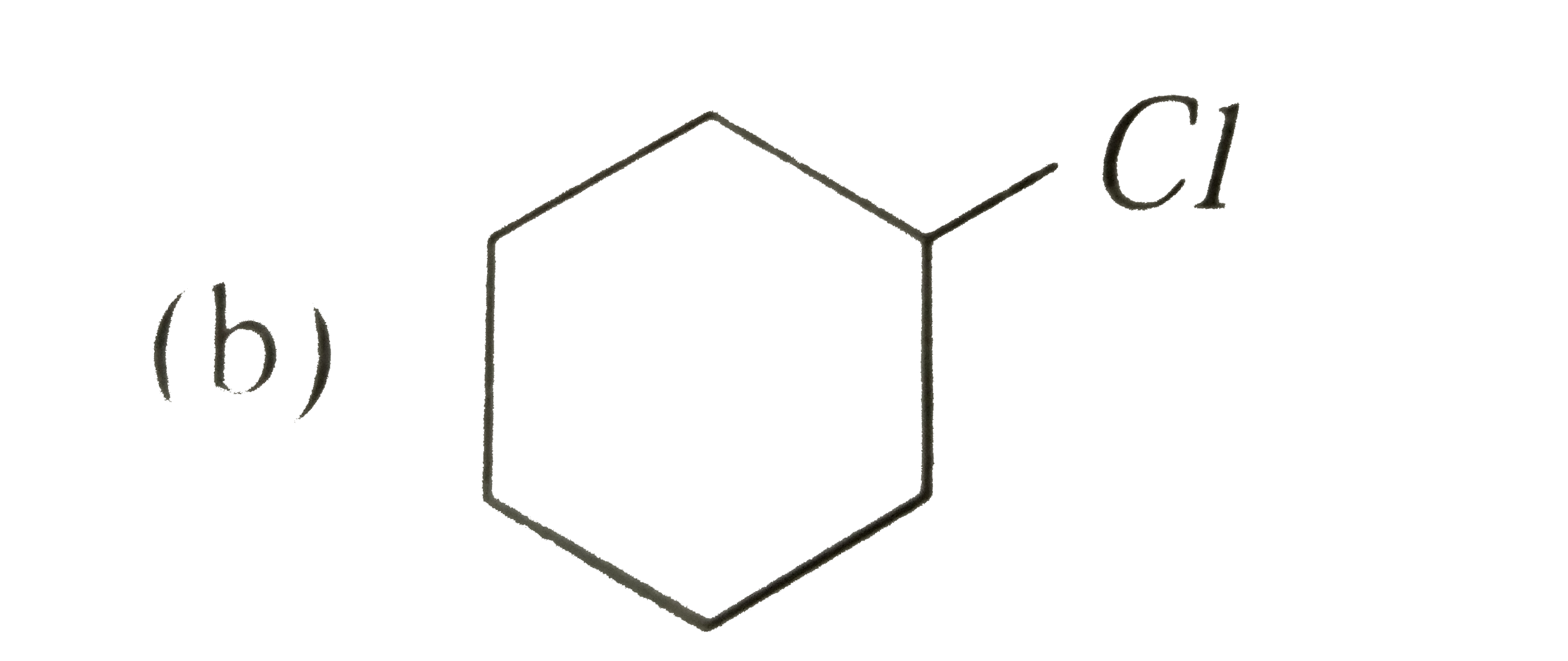
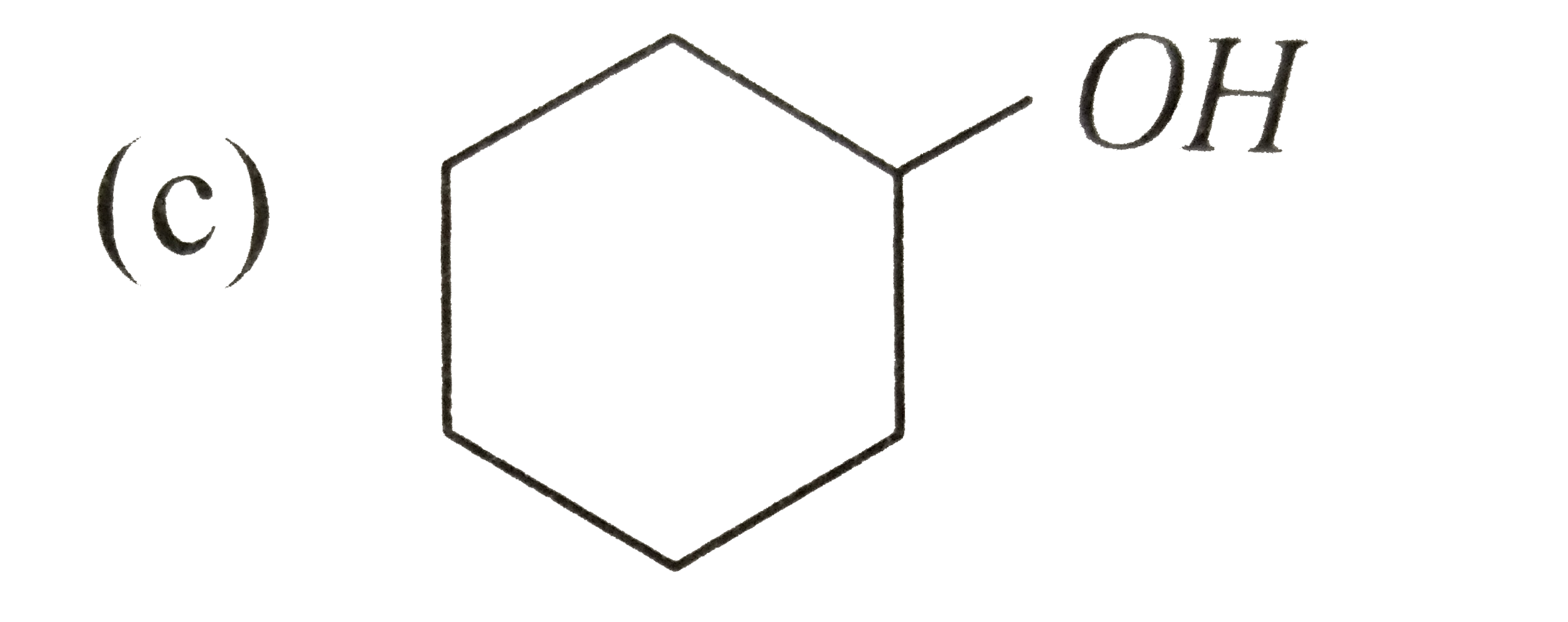
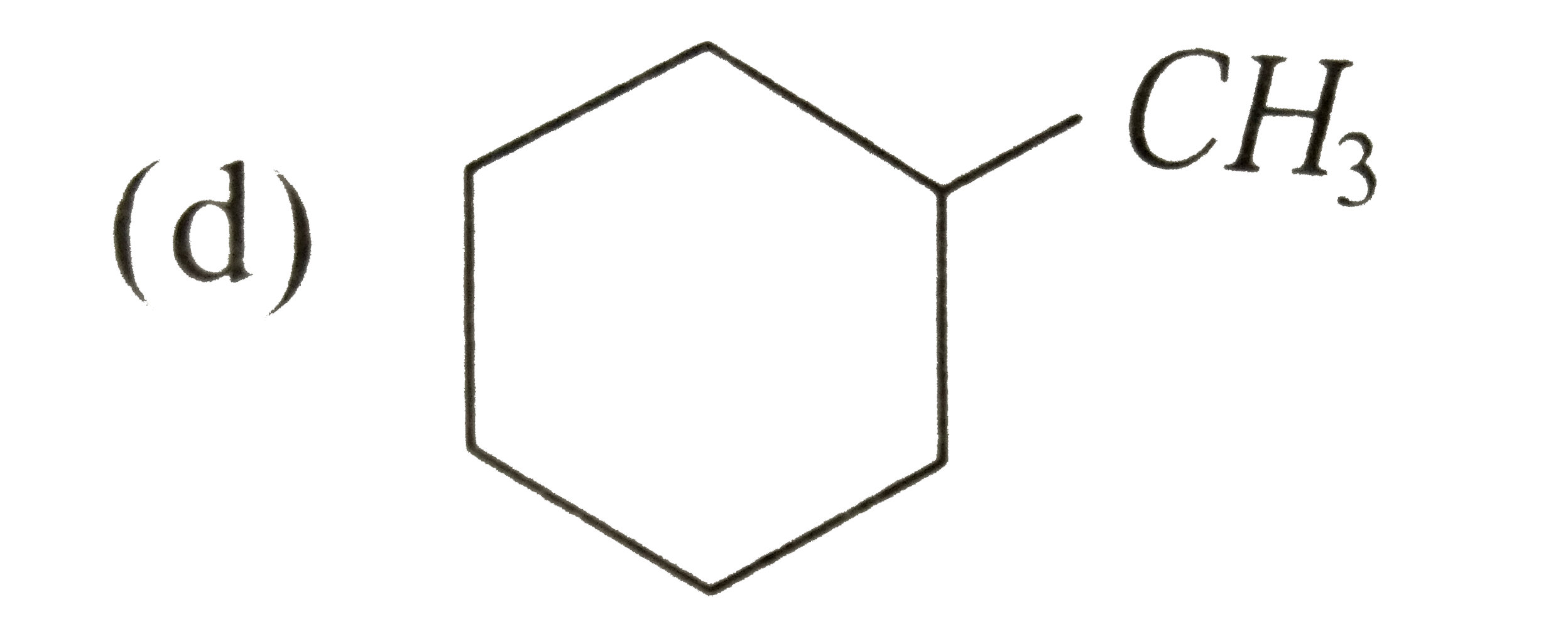
A
B
C
D
Text Solution
AI Generated Solution
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- Which of the following compound will be most easily attacked by an ele...
Text Solution
|
- Which one of the following will be the most easiliy attacked by an ele...
Text Solution
|
- Which of the following is the most reactive towards attack by an elect...
Text Solution
|
- Which of the following is the most reactive towards electrophilic atta...
Text Solution
|
- Which one of the following will be the most easiliy attacked by an ele...
Text Solution
|
- Which of the following compound will be most easily attacked by an ele...
Text Solution
|
- Which of the following compounds will be most easily attacked by an el...
Text Solution
|
- Which of the following will be most easily attacked by an electrophili...
Text Solution
|
- The compound which is most reactiv in case of electrophilic attack is
Text Solution
|