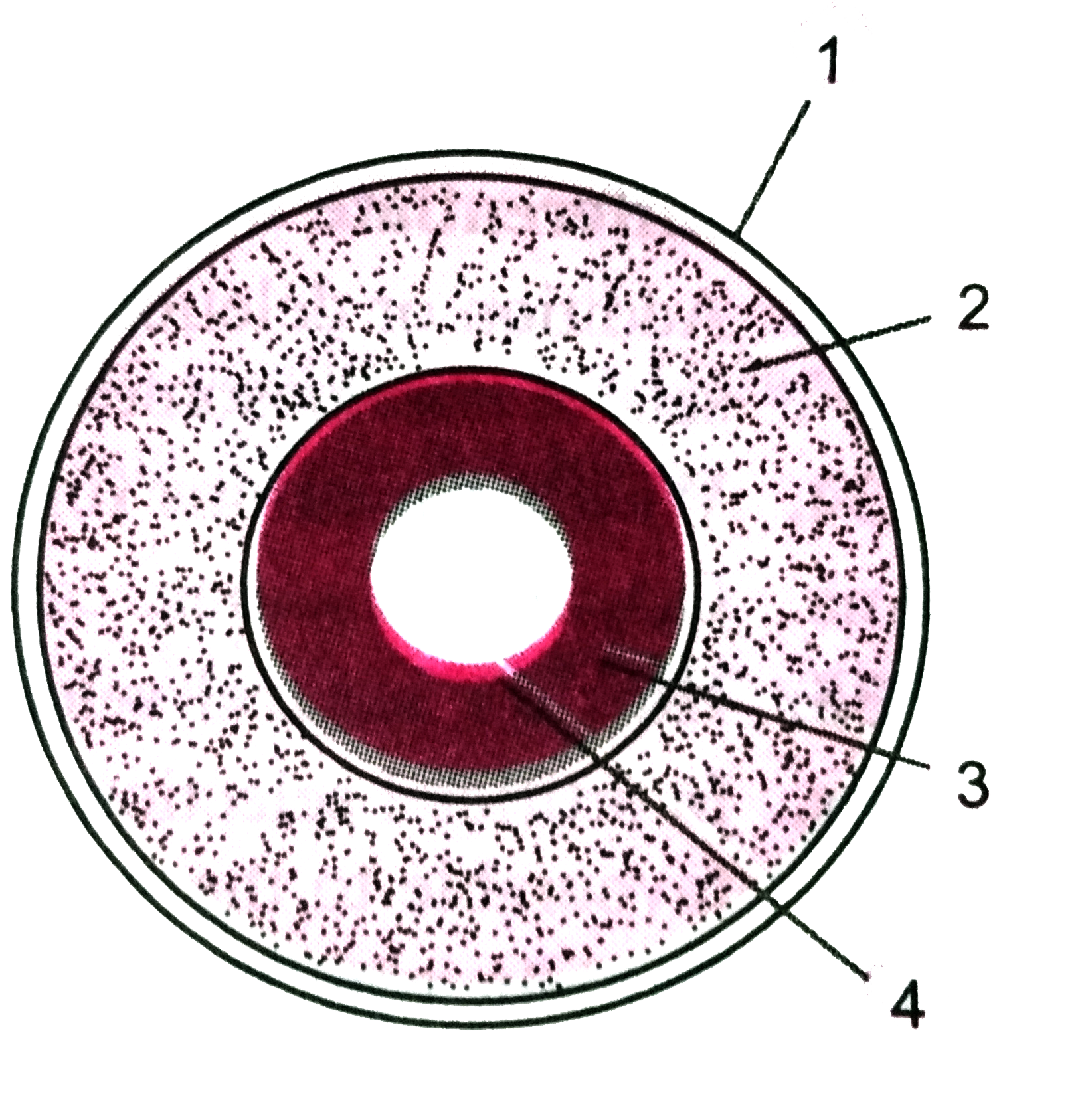
Text Solution
Verified by Experts
Similar Questions
Explore conceptually related problems
Recommended Questions
- Diagram of Earth is given . Answer the question : (a)Label the regio...
Text Solution
|
- Which option is correct for the region labelled as a, b, c and d in th...
Text Solution
|
- Name the regions marked A,B and C
Text Solution
|
- (a) Write the names of the regions in hinbrain. Give one function of e...
Text Solution
|
- Which region is considered as the one with highest biodiversity on ear...
Text Solution
|
- Name the important industrial regions of India.
Text Solution
|
- Observe the map and answer the questions that follow: Identify the i...
Text Solution
|
- Name the regions marked A,B and C
Text Solution
|
- Name the regions marked A, B and C.
Text Solution
|