Text Solution
Verified by Experts
Topper's Solved these Questions
THE P-BLOCK ELEMENTS
AAKASH INSTITUTE|Exercise Assignment Section-A)|94 VideosTHE P-BLOCK ELEMENTS
AAKASH INSTITUTE|Exercise Assignment Section-B)|26 VideosTHE P-BLOCK ELEMENTS
AAKASH INSTITUTE|Exercise Assignment Section-J)|16 VideosTHE D AND F-BLOCK ELEMENTS
AAKASH INSTITUTE|Exercise Assingment ( Section-J Aakash Challengers Questions )|10 VideosTHE S-BLOCK ELEMENTS
AAKASH INSTITUTE|Exercise Assignment (Section-J)|10 Videos
Similar Questions
Explore conceptually related problems
AAKASH INSTITUTE-THE P-BLOCK ELEMENTS-Try yourself
- Complete the following reactions : (i) C(2)H(4)+O(2) to (ii) Al +O...
Text Solution
|
- Why does O(3) act as a powerful oxidising agent ?
Text Solution
|
- How is O(3) estimated quantitatively ?
Text Solution
|
- What happens when sulphur dioxide is passed through an aqueous solutio...
Text Solution
|
- Comment on the nature of two S-O bonds formed in SO(2) molecule. Are t...
Text Solution
|
- How is the presence of SO2 detected?
Text Solution
|
- Mention three areas in which H(2)SO(4) plays an important role.
Text Solution
|
- Write the conditions to maximize the yield of H(2)SO(4) by contact pro...
Text Solution
|
- Why is K(a(2)) ltlt K(a(1)) for H(2)SO(4) in water?
Text Solution
|
- Considering the parameters such as bond dissociation enthalpy, electro...
Text Solution
|
- Give two examples to show the anomalous behaviour of fluorine.
Text Solution
|
- Give two examples to show the anomalous behaviour of fluorine.
Text Solution
|
- Sea is the greatest source of some halogens. Comment.
Text Solution
|
- Name three gases which can be prepared from chlorine gas.
Text Solution
|
- Why is ICl more reactive than I(2) ?
Text Solution
|
- Draw structure and give hybridisation of IF(7).
Text Solution
|
- Why is helium used in diving apparatus ?
Text Solution
|
- Balance the equation : XeF(6)+H(2)O to XeO(2)F(2)+HF
Text Solution
|
- Why has it been difficult to study the chemistry of radon ?
Text Solution
|
- List the uses of neon and argon gases.
Text Solution
|
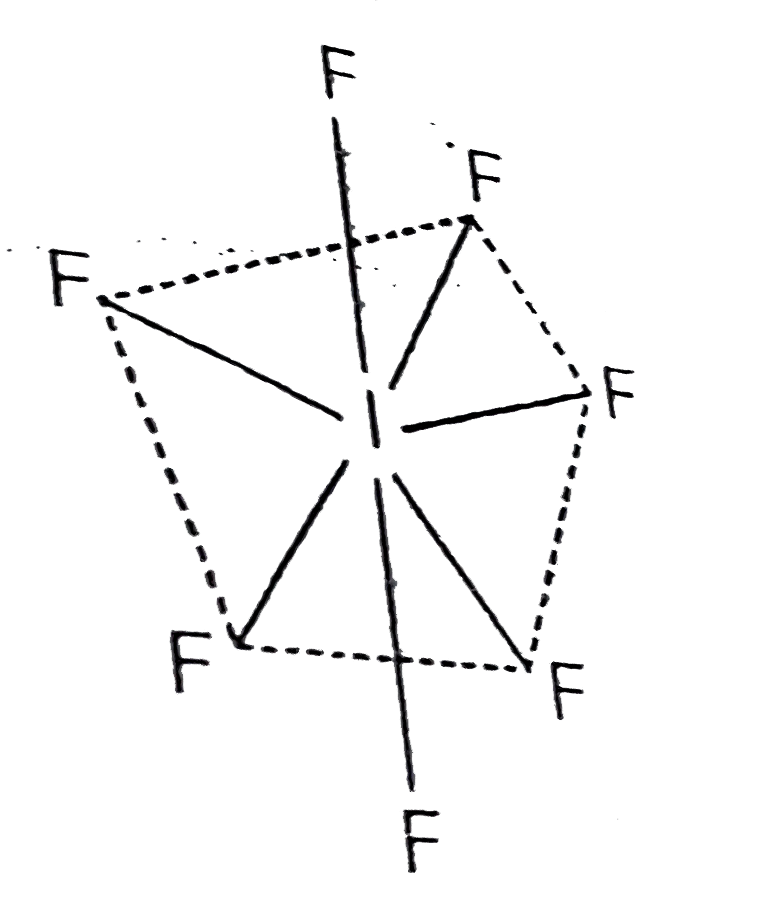 .
.