Similar Questions
Explore conceptually related problems
Recommended Questions
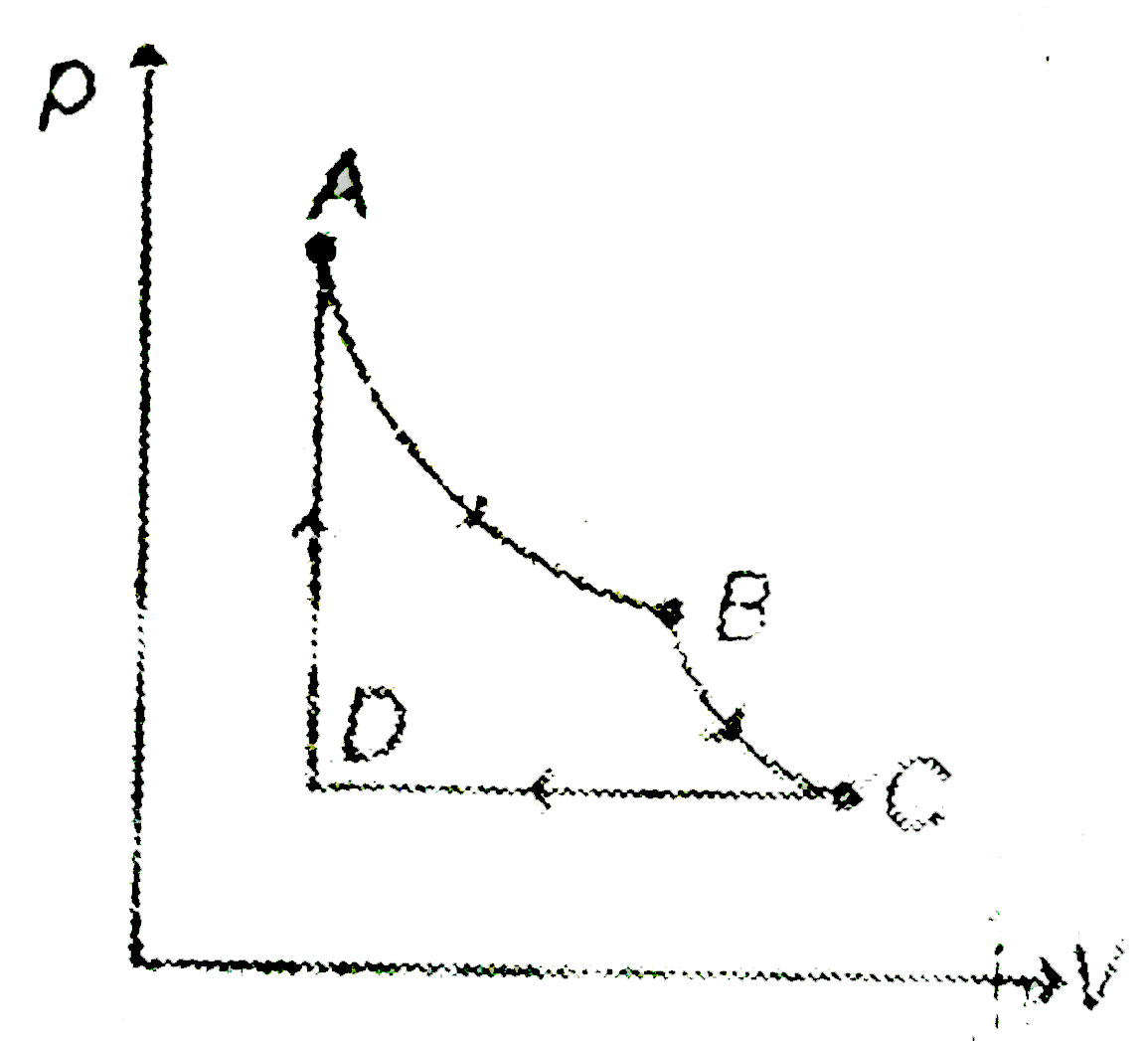
- Two moles of a diatomic gas are carried trhough the cycle ABCDA as sho...
Text Solution
|
- If a certain mass of gas is made to undergo separately adiabatic and i...
Text Solution
|
- Two moles of a diatomic gas are carried trhough the cycle ABCDA as sho...
Text Solution
|
- Two moles of a diatomic gas are carried trhough the cycle ABCDA as sho...
Text Solution
|
- Two moles of a diatomic gas are carried trhough the cycle ABCDA as sho...
Text Solution
|
- One mole of oxygen of volume 1 litre at 4 atm pressure to attain 1 atm...
Text Solution
|
- One mol of an ideal diatomic gas underwent an adiabatic expansion form...
Text Solution
|
- A gas has an initial volume of 1L at a pressure of 8 atm. An adiabatic...
Text Solution
|
- The initial volume of a gas is 1 L and the pressure is 16 atm. Due to ...
Text Solution
|