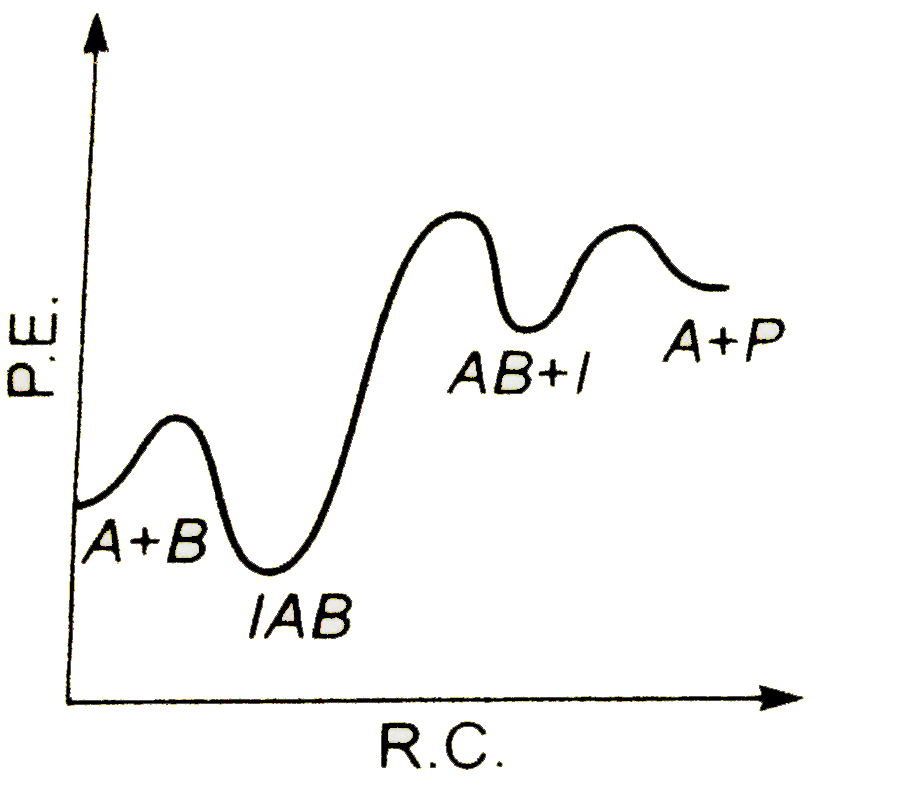
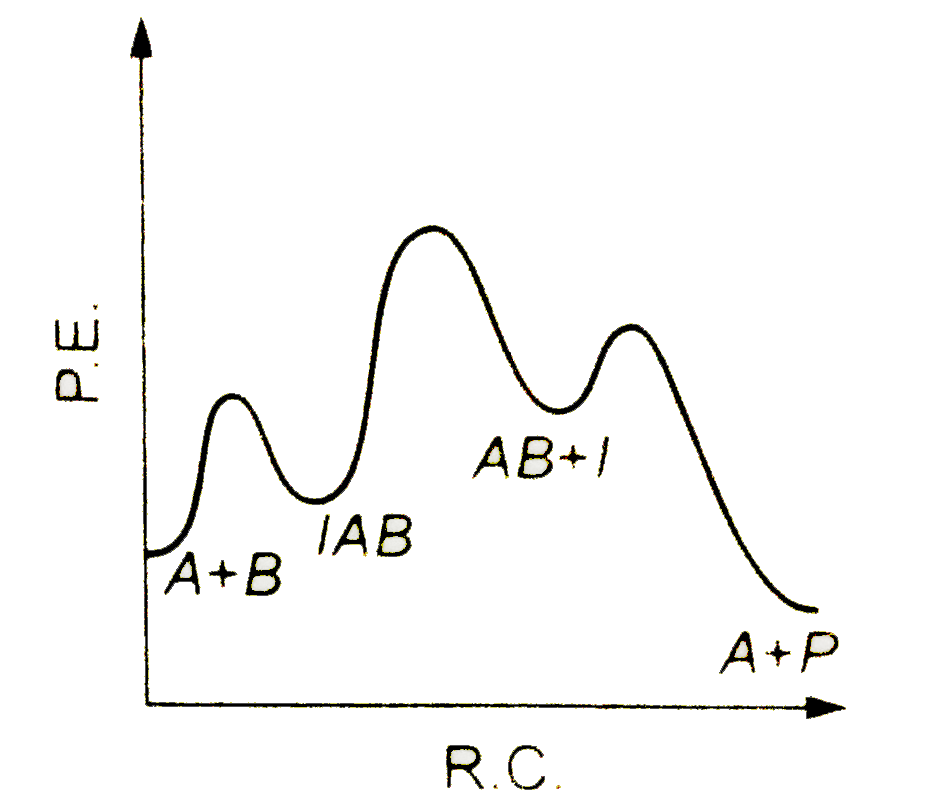
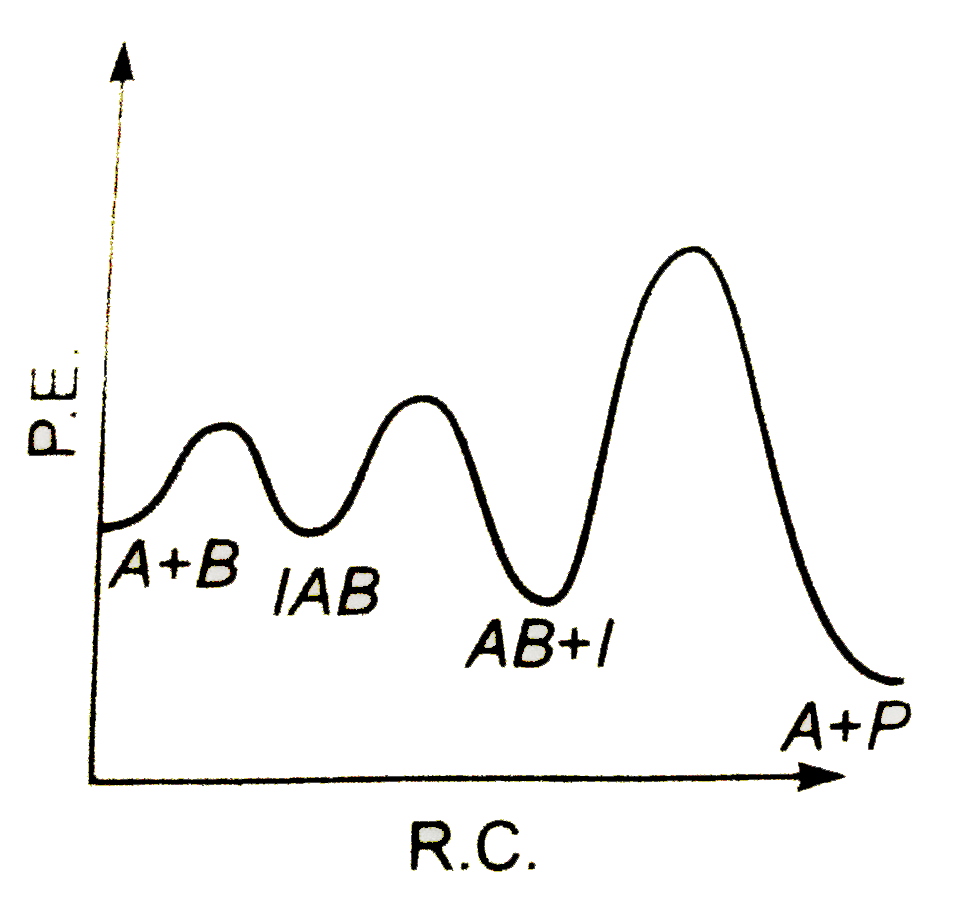
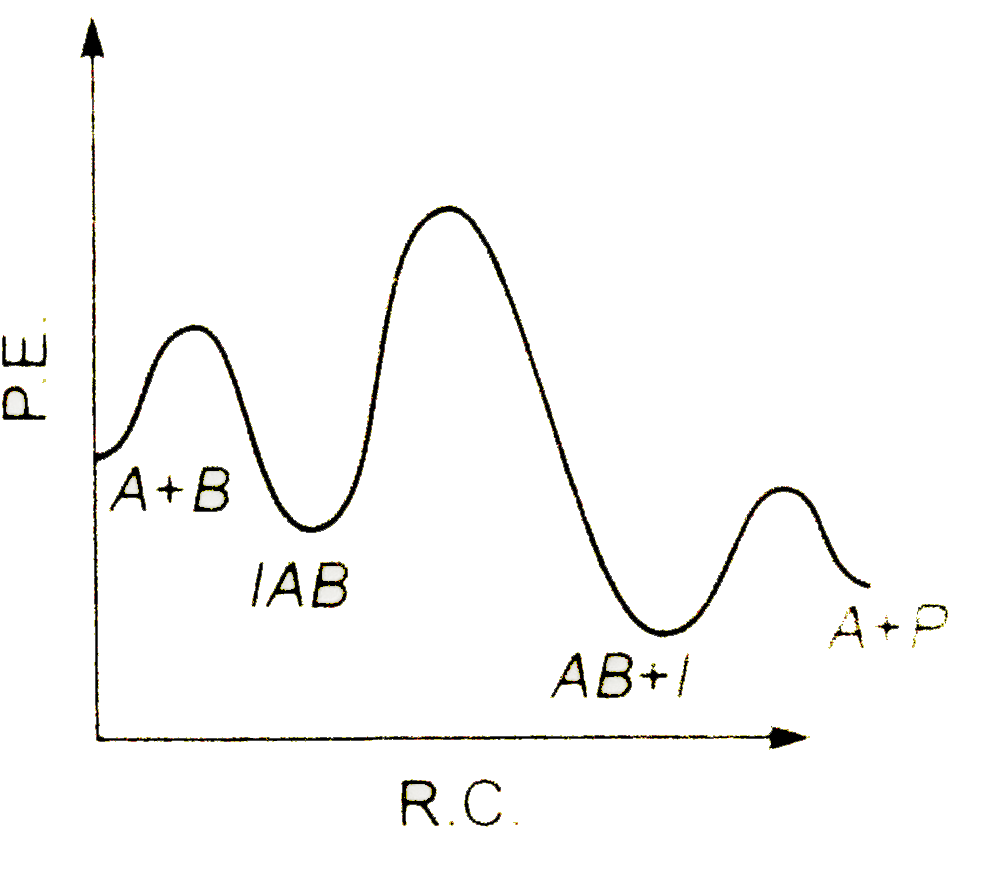
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- The following mechanism has been proposed for the exothermic catalyzed...
Text Solution
|
- Mechanism of the reaction is: A overset(k(1))rarrB, 2Aoverset(k(2))ra...
Text Solution
|
- Conisder the reaction mechanism: A(2) overset(k(eq))hArr 2A ("fast") (...
Text Solution
|
- The following mechanism has been proposed for the exothermic catalyzed...
Text Solution
|
- The mechanism of the reaction 2NO + O(2) rarr 2NO(2) is NO + NO un...
Text Solution
|
- A consecutive reaction, A overset(K(1))(rarr)B overset(K(2))(rarr)C is...
Text Solution
|
- A hypothetical reaction : A(2)+B(2)rarr 2AB Follows mechanism as given...
Text Solution
|
- In the sequence of reaction, L overset(k(1))rarr M overset(k(2))rar...
Text Solution
|
- In the following first order reactions (A)overset(k(1))rarr"product",(...
Text Solution
|