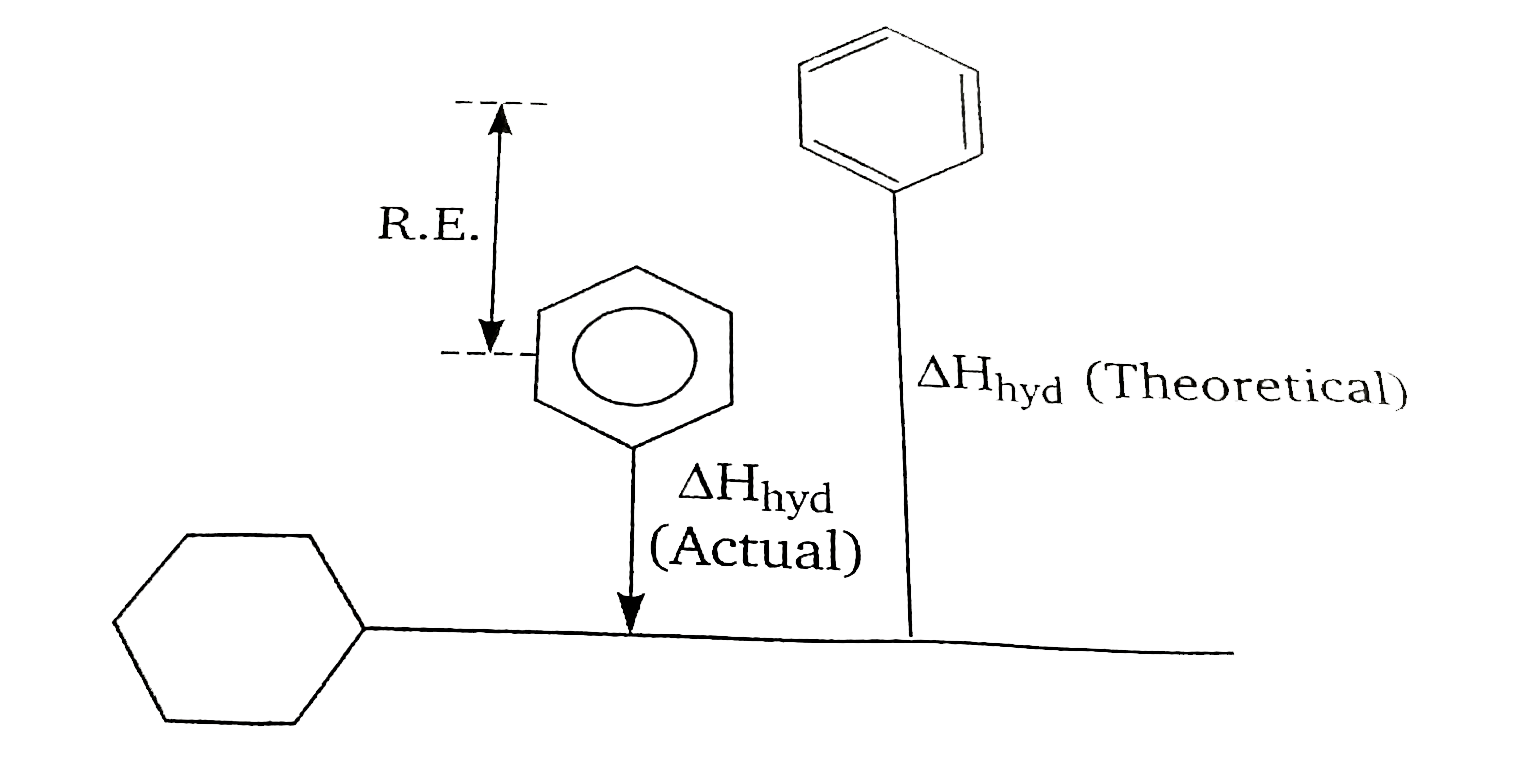
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
Similar Questions
Explore conceptually related problems
NARENDRA AWASTHI ENGLISH-THERMODYNAMICS-Level 2
- Calculate Delta(r)G^(@) for (NH(4)Cl,s) at 310K. Given :Delta(r)H^(@...
Text Solution
|
- Using listed informations, calculate Delta(r)G^(@) (in kJ/mol) at 27^(...
Text Solution
|
- Fixed mass of an ideal gas collected in a 24.64 litre sealed rigid ves...
Text Solution
|
- The molar heat capacities at constant pressure (assumed constant with ...
Text Solution
|
- Determine Delta U^(@) at 300K for the following reaction using the li...
Text Solution
|
- Calculate Delta(f)H^(@) (in kJ/mol) for Cr(2)O(3) from the Delta(r)G^(...
Text Solution
|
- Calculate the heat produced (in kJ) when 224 gm of CaO is completely c...
Text Solution
|
- When 1.0 g of oxalic acid (H(2)C(2)O(4)) is burnt in a bomb calorimete...
Text Solution
|
- Enthalpy of neutralization of H(3)PO(3) "with " NaOH " is" -106.68 "kJ...
Text Solution
|
- The enthalpy of neutralization of a Weak monoprotic acid (HA) in 1 M s...
Text Solution
|
- Determine C-C and C-H bond enthalpy (in kJ//mol) Given : Delta(f)H^(...
Text Solution
|
- Consider the following data : Delta(f)H^(@)(N(2)H(4),l)=50kJ//mol,Delt...
Text Solution
|
- What is the bond enthalpy of Xe-F bond ? XeF(4)(g)rarrXe^(+)(g)+F^(-...
Text Solution
|
- If enthaopy of hydrogenation of C(6)H(6)(l) into C(6)H(12)(l) is -205k...
Text Solution
|
- The enthalpy of combustion of propance (C(3)H(8)) gas in temes of give...
Text Solution
|
- The efficiency of an ideal gas with adiabatic exponent 'gamma' for the...
Text Solution
|
- Initially one mole of ideal gas (C(v)=(5)/(2)R) at 0.1 atm and 300 K i...
Text Solution
|
- What is the enthalpy of neutralization of HF against a strong base? ...
Text Solution
|
- The molar heat capacities of Iodine vapour and solid are 7.8 and 14 ca...
Text Solution
|
- For the reaction taking place at certain temperature NH(2)COONH(4)(s)h...
Text Solution
|