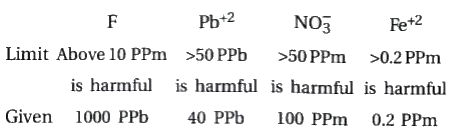A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
ENVIRONMENT CHEMISTRY
KUMAR PRAKASHAN|Exercise SECTION-D NCERT EXEMPLAR SOLUTION (Multiple Choice Questions (MCQs)|32 VideosENVIRONMENT CHEMISTRY
KUMAR PRAKASHAN|Exercise SECTION-D NCERT EXEMPLAR SOLUTION (Multiple Choice Questions (MCQs)(More than one correct answer)|1 VideosENVIRONMENT CHEMISTRY
KUMAR PRAKASHAN|Exercise SECTION-C (Multiple Choice Questions (MCQs) (MCQs asked in Board Exam)|19 VideosCLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES
KUMAR PRAKASHAN|Exercise Section -D SOLUTIONS OF NCERT EXEMPLAR PROBLEMS (Long Answer Type Questions )|7 VideosEQUILIBRIUM
KUMAR PRAKASHAN|Exercise Question Paper from Module|7 Videos
Similar Questions
Explore conceptually related problems
KUMAR PRAKASHAN-ENVIRONMENT CHEMISTRY -SECTION-C (Multiple Choice Questions (MCQs) (MCQs asked in JEE / NEET / AIEEE)
- The green house effect is because of the
Text Solution
|
- Which of the following is/are the hazardous pollutant(s) present in au...
Text Solution
|
- Green chemistry means such reactions which :
Text Solution
|
- Which one of the following statement is not true ?
Text Solution
|
- Assertion : Nitrogen and Oxygen are the main components in the atmosph...
Text Solution
|
- The concentration of fluoride, lead, nitrate and iron in a water sampl...
Text Solution
|
- The recommended concentration of fluoride ion in drinking water is up ...
Text Solution
|
- Which oxide of nitrogen is not a common pollutant introduced into the ...
Text Solution
|
- Among the following, the one that is not a green house gas is.....
Text Solution
|
- A water sample has ppm level concentration of the following metals: Fe...
Text Solution
|
- The reaction that is not involved in the ozone layer depletion mechani...
Text Solution
|
- Excessive release of CO2 into the atomosphere results in :
Text Solution
|
- Among the gases (i) – (v), the gases that cause greenhouse effect are ...
Text Solution
|