A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
Similar Questions
Explore conceptually related problems
NCERT FINGERTIPS ENGLISH-NEURAL CONTROL AND COORDINATION-All Questions
- In an animal assume that rough coat (R ) is domiant over smooth coat (...
Text Solution
|
- Which of the following is wheat fruit?
Text Solution
|
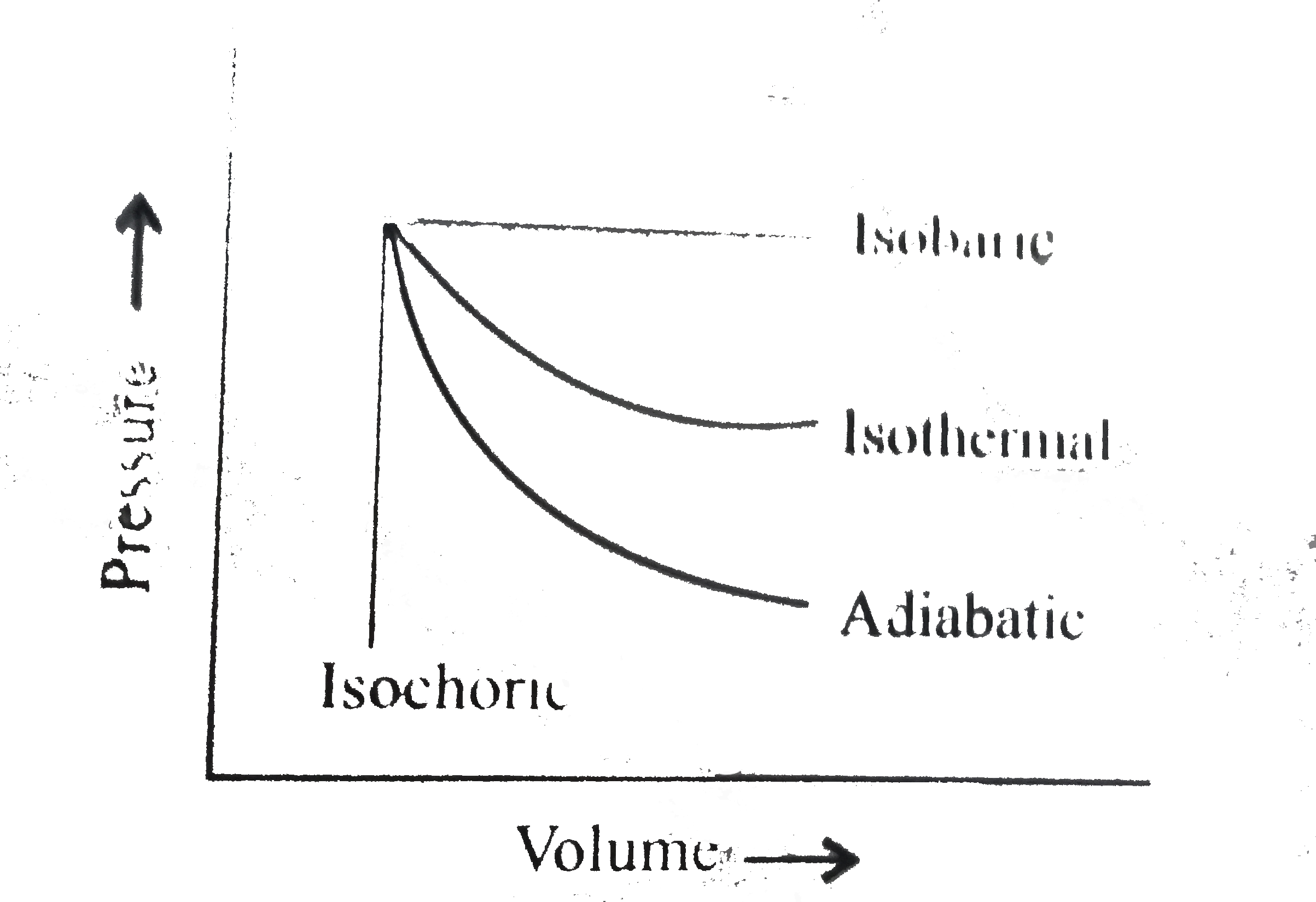
- The pressure-volume of various thermodynamic process is shown in graph...
Text Solution
|
- Pons' connects the
Text Solution
|
- Cerebellum of brain is concerned with
Text Solution
|
- Tree of life is
Text Solution
|
- Which of the following statements is correct regarding cerebellum ?
Text Solution
|
- The part of hind brain that is responsible for hand eye coordination i...
Text Solution
|
- The respiratory and cardiac centres are located in
Text Solution
|
- Salivation in mammals is under the control of
Text Solution
|
- Which labelled part controls the process of breathing ?
Text Solution
|
- Which of the following parts of brain constitute the brain stem ?
Text Solution
|
- Cell bodies of neurons bringing afferent information into the spinal c...
Text Solution
|
- Which of the following is wheat fruit?
Text Solution
|
- Mark the correct match
Text Solution
|
- For good reflex actions we require intact
Text Solution
|
- The path of reflex arc is shown in the given figure.Identify the diffe...
Text Solution
|
- Which of the following is an example of conditioned reflex ?
Text Solution
|
- Which of the following is not a relfex action ?
Text Solution
|
- The given diagrammatic representation of reflex action shows knee jerk...
Text Solution
|