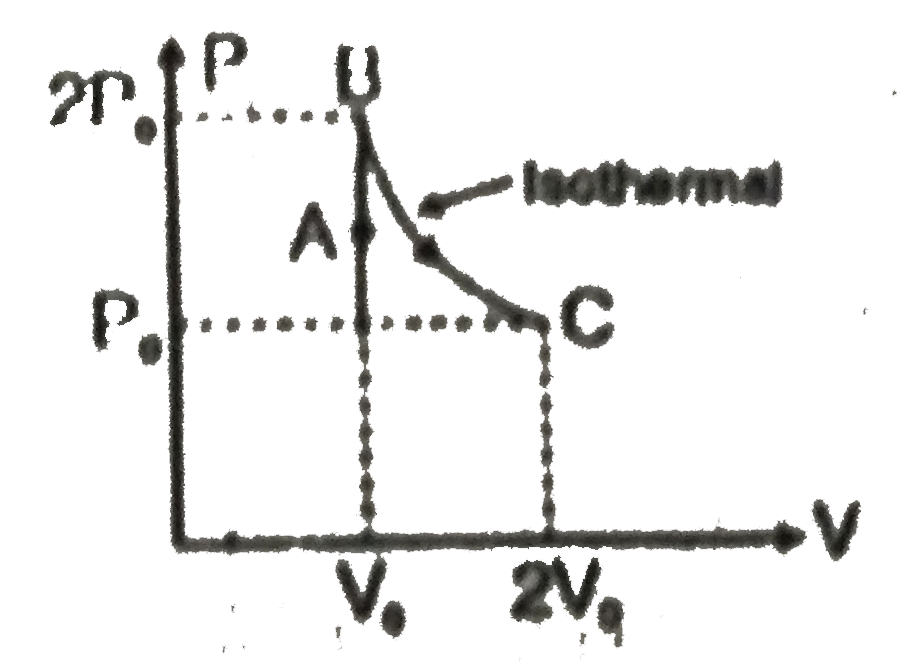
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
Similar Questions
Explore conceptually related problems
DC PANDEY ENGLISH-CURRENT ELECTRICITY-All Questions
- The resistance of a series combination of the resistance is S. when t...
Text Solution
|
- The following sets of values for C(v) and C(p) of an ideal gas have be...
Text Solution
|
- A diatomic ideal gas undergoes a thermodynamic change according to the...
Text Solution
|
- P - T diagram is shown in Fig. Choose the corresponding V - T diagram.
Text Solution
|
- A small quantity mass m, of water at a temperature theta ("in " ^(@)C)...
Text Solution
|
- Two rods having length l(1) and l(2), made of materials with the linea...
Text Solution
|
- A wall is made of equally thick layers A and B of different matierals...
Text Solution
|
- A wall consists of alternating blocks withlength 'd' and coefficient o...
Text Solution
|
- A rod of length 2m rests on smooth horizontal floor. If the rod is hea...
Text Solution
|
- Steam at 100^(@)C is added slowly to 1400 gm of water at 16^(@)C until...
Text Solution
|
- A block of ice with mass m falls into a lake. After impact, a mass of ...
Text Solution
|
- The specific heat of a metal at low temperatures varies according to S...
Text Solution
|
- Steam at 100^(@)C is passed into 1.1 kg of water contained in a calori...
Text Solution
|
- Two sheets of thickness d and 2d and same area are touching each other...
Text Solution
|
- A composite rod made of three rods of equal length and cross-section a...
Text Solution
|
- A black metal foil is warmed by radiation from a small sphere at tempe...
Text Solution
|
- Two perfect monoatomic gases at absolute temperature T(1) and T(2) are...
Text Solution
|
- For a monoatomic ideal gas undergoing an adiabatic change, the relatio...
Text Solution
|
- A black body calorimeter filled with hot water cools from 60^(@)C to 5...
Text Solution
|
- In the process PV = constant, pressure (P) versus density (rho) graph ...
Text Solution
|