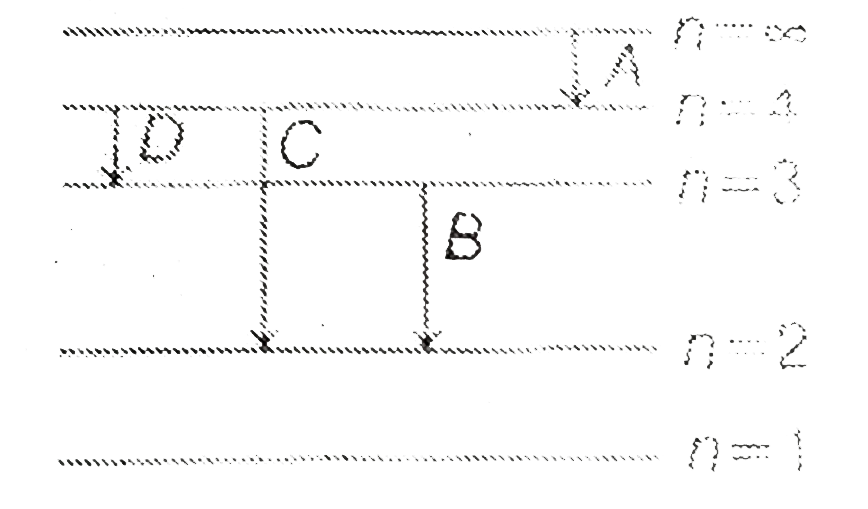A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
MODERN PHYSICS
DC PANDEY ENGLISH|Exercise for JEE Advanced (only one option is Correct)|76 VideosMODERN PHYSICS
DC PANDEY ENGLISH|Exercise for JEE Advanced (More than One Options is Correct )|1 VideosMAGNETISM AND MATTER
DC PANDEY ENGLISH|Exercise Medical gallery|1 VideosMODERN PHYSICS - 1
DC PANDEY ENGLISH|Exercise Level 2 Subjective|23 Videos
Similar Questions
Explore conceptually related problems
DC PANDEY ENGLISH-MODERN PHYSICS-Integer Type Questions
- Consider the electron energy level diagram of H-atom Photons associate...
Text Solution
|
- The ratio between acceleration of the electron in singlely ionized hel...
Text Solution
|
- If the radius of firs Bohr's orbit is x, then de-Broglie wavelenght of...
Text Solution
|
- A H-atom moving with speed v makes a head on collisioon with a H-atom ...
Text Solution
|
- The magnetic fieold at the centre of a hydrogen atom due to the motion...
Text Solution
|
- when the voltage applied to an X-ray tube increases from V(1) = 10 kV ...
Text Solution
|
- Light of wavelength 330nm falling on a piece of metal ejects electrons...
Text Solution
|
- Light of wavelength 0.6mum from a sodium lamp falls on a photocell and...
Text Solution
|
- An alpha-particle accelerated through V volt is fired towards a nucleu...
Text Solution
|
- The kinetic energies of the photoelectrons ejected from a metal surfac...
Text Solution
|
- In a hydrogen atom following the Bohr's psotulates the product of line...
Text Solution
|
- A target element A is bombarded with electrons and the wavelengths of ...
Text Solution
|
- A moving hydrogen atom makes a head on collision with a stationary hy...
Text Solution
|
- A radioactive sample contains two different types of radioactive nucle...
Text Solution
|
- A overset(lambda)rarr B overset(2 lambda)rarr C T=0 ,N(0) , 0 , T...
Text Solution
|
- In a photeclectric experiment , when electromegnetic wave given by E=E...
Text Solution
|
- Nuclei X and Y convert into a stable nucleus Z. At t=0 , the number o...
Text Solution
|
- Single electron is orbiting in n^(th)orbit of hydrogen atom. The magne...
Text Solution
|