Topper's Solved these Questions
Similar Questions
Explore conceptually related problems
MAXIMUM PUBLICATION-MODEL PAPER 1-EXAMPLE
- The electronic configuration of some elements are given below. 12A=2...
Text Solution
|
- The electronic configuration of some elements are given below. 12A=2...
Text Solution
|
- Match the following suitably
Text Solution
|
- Find out the oxidation number of Mn in the Following compounds Hint:...
Text Solution
|
- Find out the oxidation number of Mn in the Following compounds Hint:...
Text Solution
|
- Find out the oxidation number of Mn in the Following compounds Hint:...
Text Solution
|
- The symbol of these atoms are given below. 6C^12, 7N^14, 6C^14 Whi...
Text Solution
|
- The symbol of these atoms are given below. 6C^12, 7N^14, 6C^14 Wha...
Text Solution
|
- The symbol of these atoms are given below. 6C^12, 7N^14, 6C^14 Wri...
Text Solution
|
- Compare the properties of ionic and covalent compounds.
Text Solution
|
- The symbol of certain elements are given. Write their electronic confi...
Text Solution
|
- The Bohr model of an atom is given below. Analyse it and find out the ...
Text Solution
|
- The Bohr model of an atom is given below. Analyse it and find out the ...
Text Solution
|
- The Bohr model of an atom is given below. Analyse it and find out the ...
Text Solution
|
- The Bohr model of an atom is given below. Analyse it and find out the ...
Text Solution
|
- The electronegativity values of two elements are given below. (C=2.55 ...
Text Solution
|
- The electronegativity values of two elements are given below. (C=2.55 ...
Text Solution
|
- What is the relationship between the rate of chemical reaction and tem...
Text Solution
|
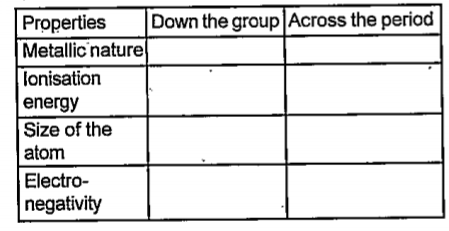
- Fill in the blanks in the following table showing how the properties v...
Text Solution
|
- Write any two advantages and two limitations of the Mendeleav's period...
Text Solution
|