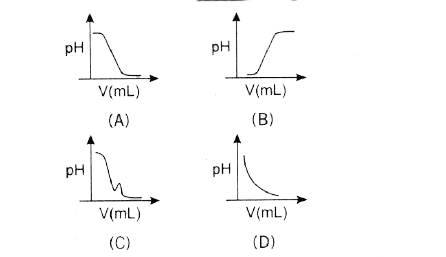
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
EQUILIBRIUM
ICSE|Exercise TRUE OR FALSE TYPE QUESTION |22 VideosEQUILIBRIUM
ICSE|Exercise FILL IN THE BLANKS TYPE QUESTIONS |34 VideosEQUILIBRIUM
ICSE|Exercise ESSAY (LONG ANSWER) TYPE QUESTIONS |17 VideosENVIRONMENTAL CHEMISTRY
ICSE|Exercise NCERT TEXT-BOOK EXERCISE|20 VideosHYDROCARBONS
ICSE|Exercise NCERT TEXT-BOOK EXERCISES |46 Videos
Similar Questions
Explore conceptually related problems
ICSE-EQUILIBRIUM -OBJECTIVE (MULTIPLE CHOICE ) TYPE QUESTIONS
- A 20 litre container at 400 K contains CO2(g) at pressure 0.4 atm a...
Text Solution
|
- The equilibrium constant of the following are : " ...
Text Solution
|
- pK(a) of a weak acid (HA) and pK(b) of a weak base (BOH) are 3.2 and 3...
Text Solution
|
- The solubility of BaSO4 in water is 2.42 xx 10^(-3) gL^(-1) at 298 ...
Text Solution
|
- Which one of the following conditions will favour maximum formation o...
Text Solution
|
- Following solutions were prepared by mixing different volumes of NaOH ...
Text Solution
|
- An aqueous solution contains 0.10 MH2 S and 0.20 M HCl if the equil...
Text Solution
|
- An aqueous solution contains an unknown concentration Ba^(2+) . Wh...
Text Solution
|
- Which of the following are Lewis acids?
Text Solution
|
- Which of the following salts is the most basic in aqueous solution? ...
Text Solution
|
- An alkali is titrated against an acid with methyl orange as indicato...
Text Solution
|
- pH of a saturated solution of Ca(OH)2 is 9. The solubility product (...
Text Solution
|
- Conjugate base the Bronsted acids H2O and HF are :
Text Solution
|
- Which will make basic buffer? a) 100 m L o f 0.1 M HCl + 200 m...
Text Solution
|
- Solubility of Cd(OH)(2) in pure water is 1.84xx10^(-5)"mole"//L Calcul...
Text Solution
|
- In which one of the following equilibrium , Kp ne Kc ?
Text Solution
|
- In an acid- base titration, 0.1 M HCl solution was added to the NaO...
Text Solution
|
- If solubility product of Zr(3)(PO(4))(4) is denoted by S, then which o...
Text Solution
|
- For the solution of the gases w,x,y and z in water at 298 K the Henry'...
Text Solution
|
- S(s)+O(2)hArrSO(2)(g)" "K(1)=10^(52) 2S(s)+3O(2)hArr2SO(3)(g)" "...
Text Solution
|