Text Solution
Verified by Experts
Topper's Solved these Questions
Similar Questions
Explore conceptually related problems
PATHFINDER-P-BLOCK ELEMENTS -QUESTION BANK
- Explain the following "The ppi-ppi back bonding occurs in the halide...
Text Solution
|
- Compound (X) on reduction with LiAlH4 gives a hydride (Y) containing 2...
Text Solution
|
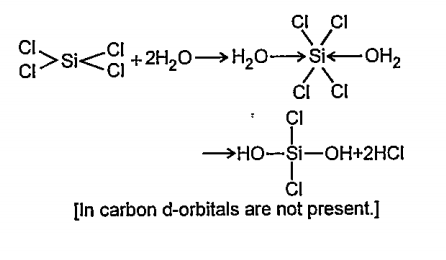
- Carbon tetrachloride is not affected but silicon tetrachloride is hydr...
Text Solution
|
- Why Sn (II) is a reducing agent whereas Pb (II) is not ?
Text Solution
|
- Graphite is a conductor but diamond is not a conductor. Explan.
Text Solution
|
- PbCl4 exist while PbBr4 and PbI4 do not exist.-Explain.
Text Solution
|
- Borax structure contains
Text Solution
|
- When orthoboric acid is heated strongly, it gives which of the followi...
Text Solution
|
- Which compounds gives O2 on moderate heating ?
Text Solution
|
- Amorphous B on heating in air forms
Text Solution
|
- AlCl3 fumes in moist air because of
Text Solution
|
- The dissolution of Al(OH)3 by a solution of NaOH results in the format...
Text Solution
|
- Boron carbide B4C is widely used in
Text Solution
|
- Which of the following compounds is optically active ?
Text Solution
|
- When a solution of NaOH is added in excess to the solution of potash a...
Text Solution
|
- Inorganic graphite is
Text Solution
|
- Which is the correct basicity order :
Text Solution
|
- R3SiCl on hydrolysis forms
Text Solution
|
- What is water glass
Text Solution
|
- Carborundum is obtained when silica is heated at high temperature with
Text Solution
|