Text Solution
Verified by Experts
Topper's Solved these Questions
SOLID STATE
CENGAGE CHEMISTRY ENGLISH|Exercise Solved Examples|13 VideosSOLID STATE
CENGAGE CHEMISTRY ENGLISH|Exercise Exercises (Linked Comprehension)|13 VideosREDUCTION AND OXIDATION REACTION OF ORGANIC COMPOUNDS
CENGAGE CHEMISTRY ENGLISH|Exercise SUBJECTIVE TYPE|4 VideosSOLUTIONS
CENGAGE CHEMISTRY ENGLISH|Exercise Ex 2.3 (Objective)|9 Videos
Similar Questions
Explore conceptually related problems
CENGAGE CHEMISTRY ENGLISH-SOLID STATE-Ex 1.2 (Objective)
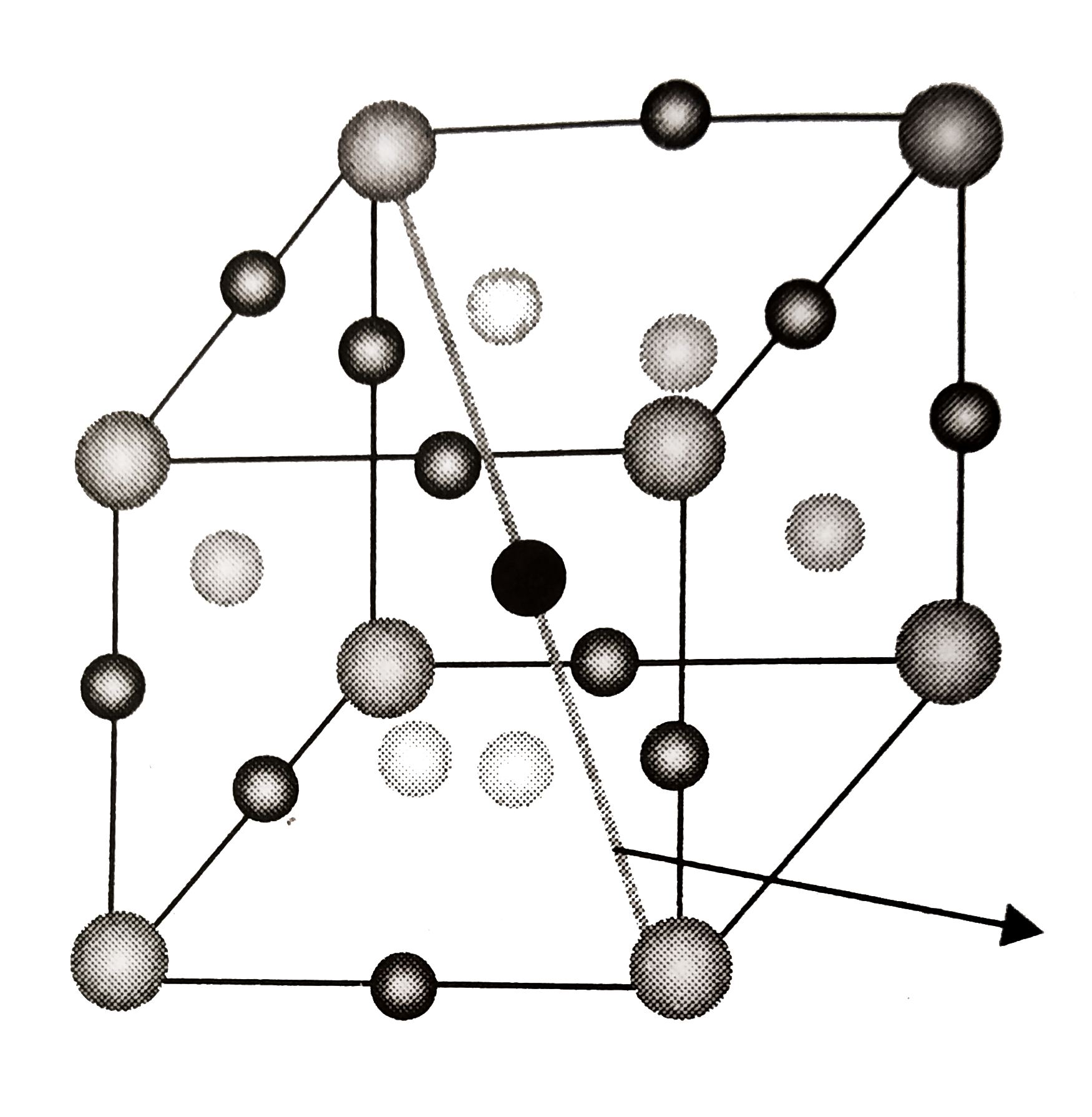
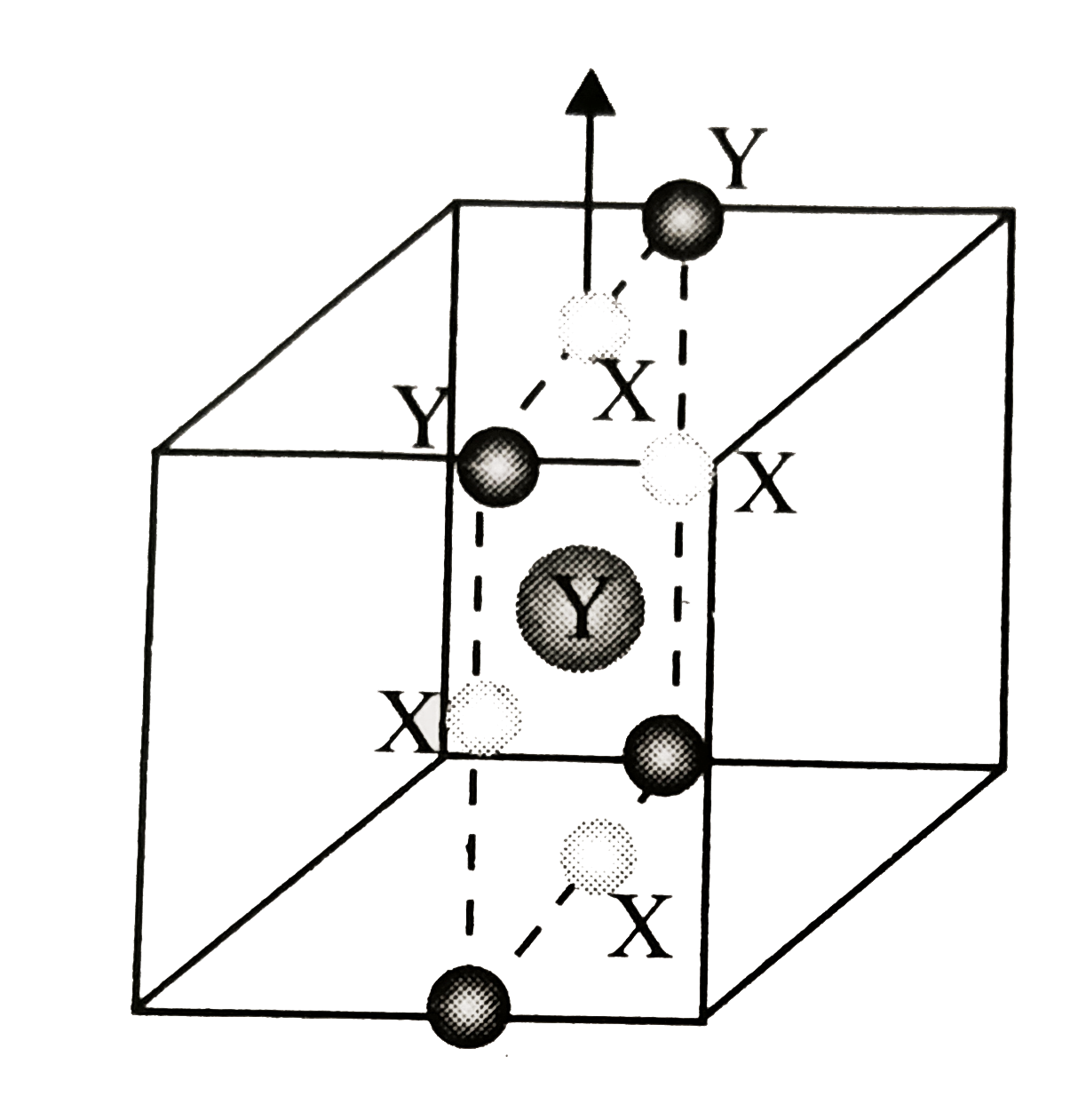
- A compound formed by elements X and Y has a cubic structure in which X...
Text Solution
|
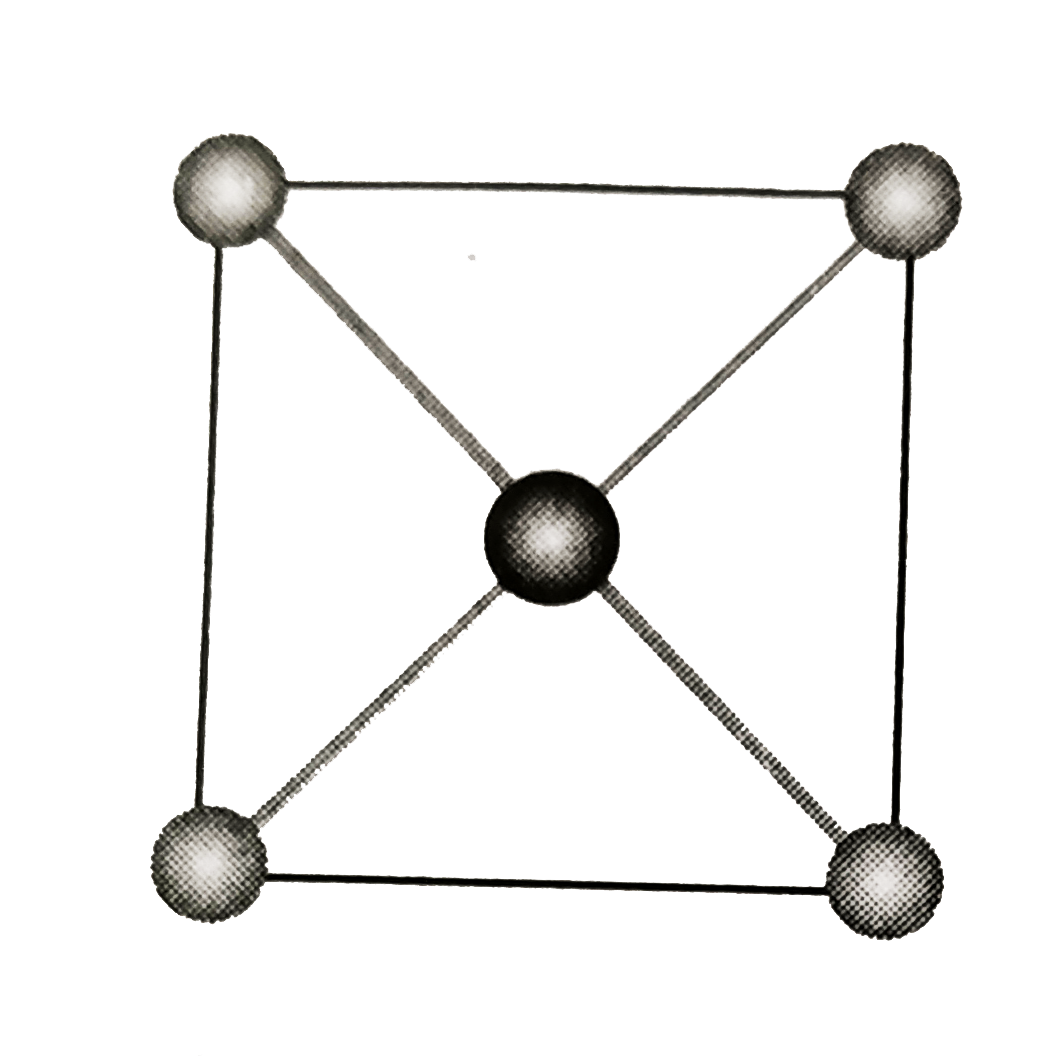
- The structure shown here represents
Text Solution
|
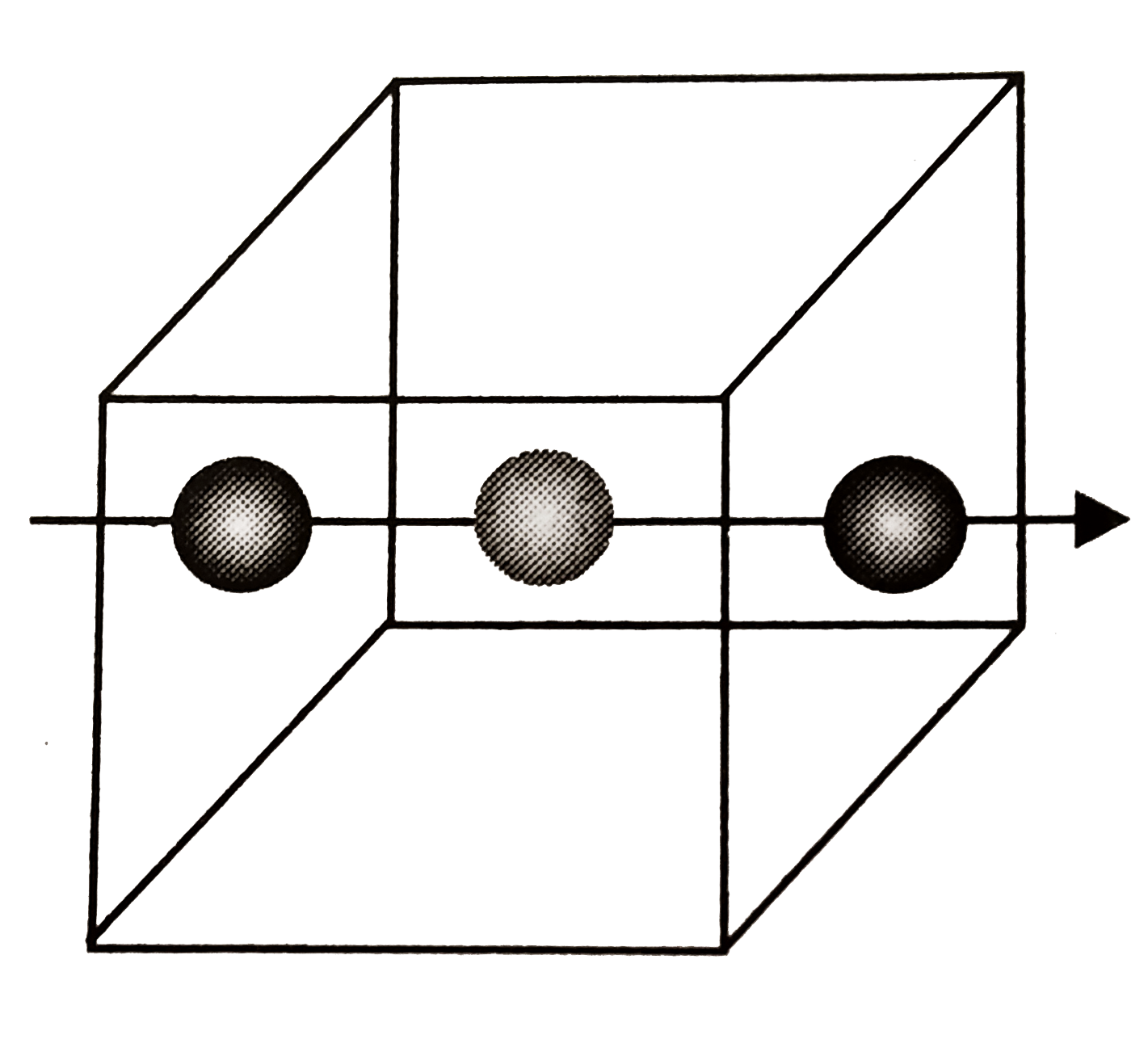
- The structure shown here represents
Text Solution
|
- In AgCl, the Ag^(o+) ions are deisplaced from their lattice position t...
Text Solution
|
- NaCl shows Schottku defects and AgCl Frenkel defects. The electrical c...
Text Solution
|
- Amorphous solids are
Text Solution
|
- Due to Frenkel defect, the density of ionic solids:
Text Solution
|
- Which of the following statement is/are correct?
Text Solution
|
- Which of the following statement is//are correct?
Text Solution
|
- Identify the Acidic salt? NaCl,NaNO3,NH4Cl
Text Solution
|