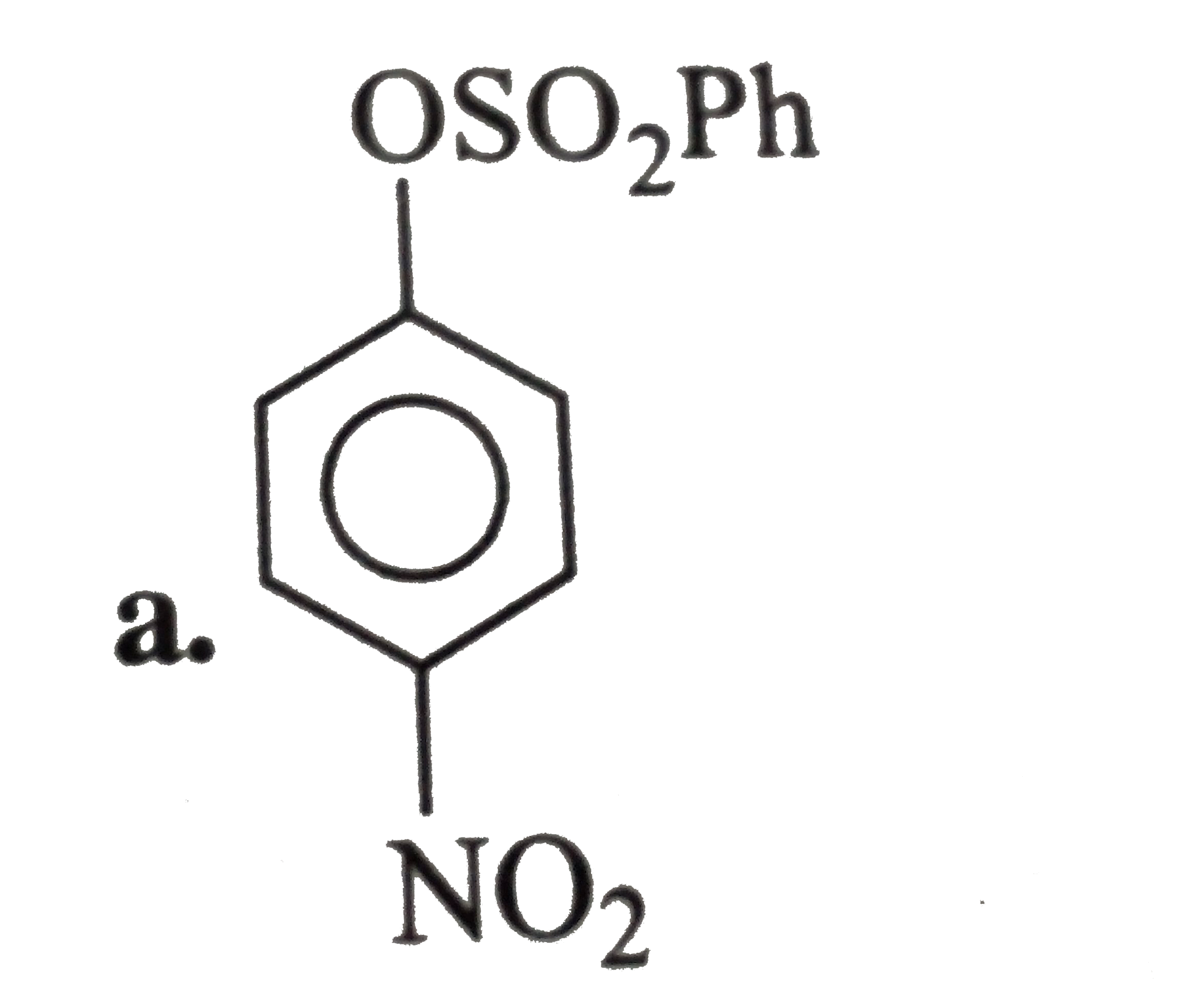
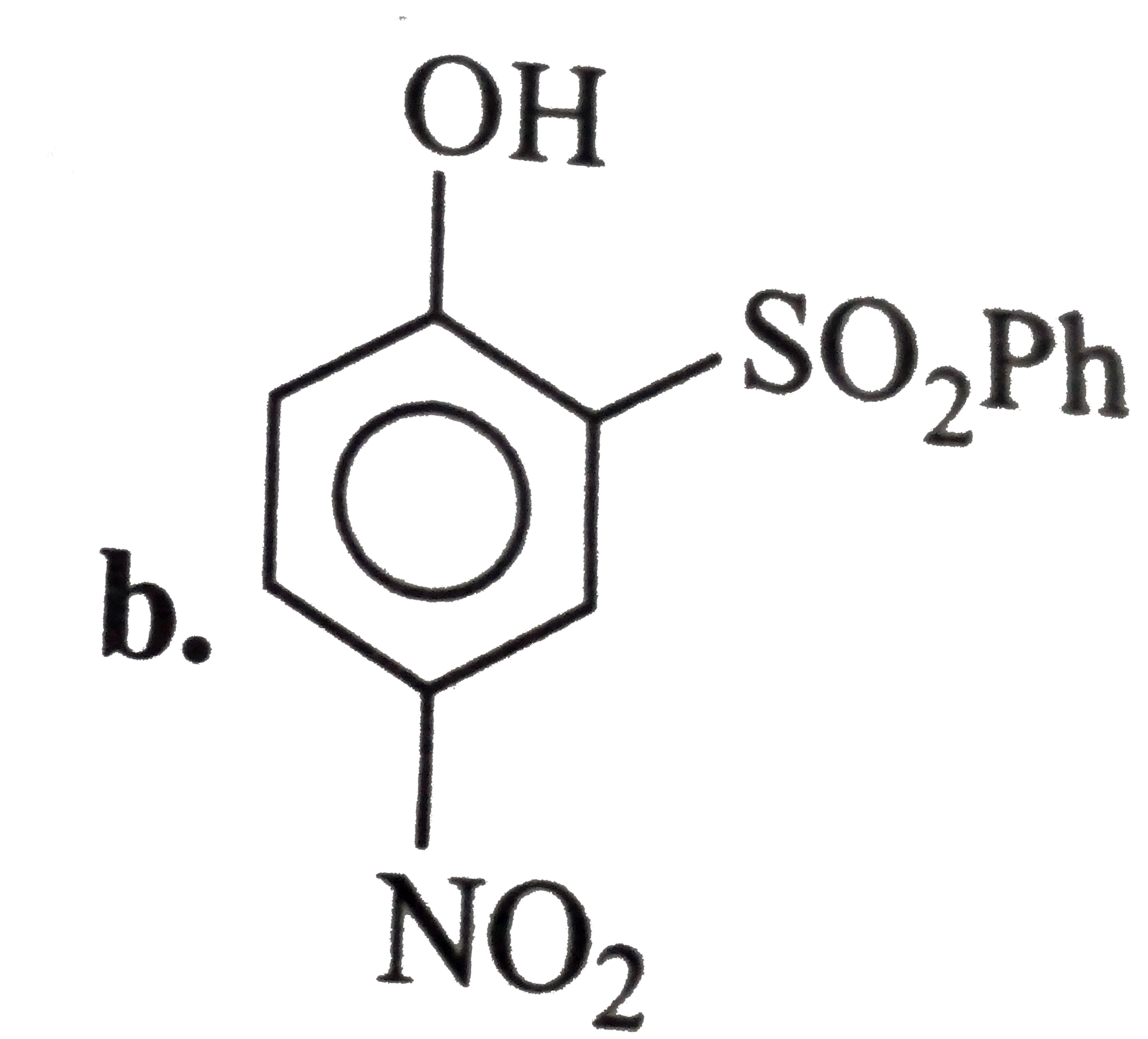
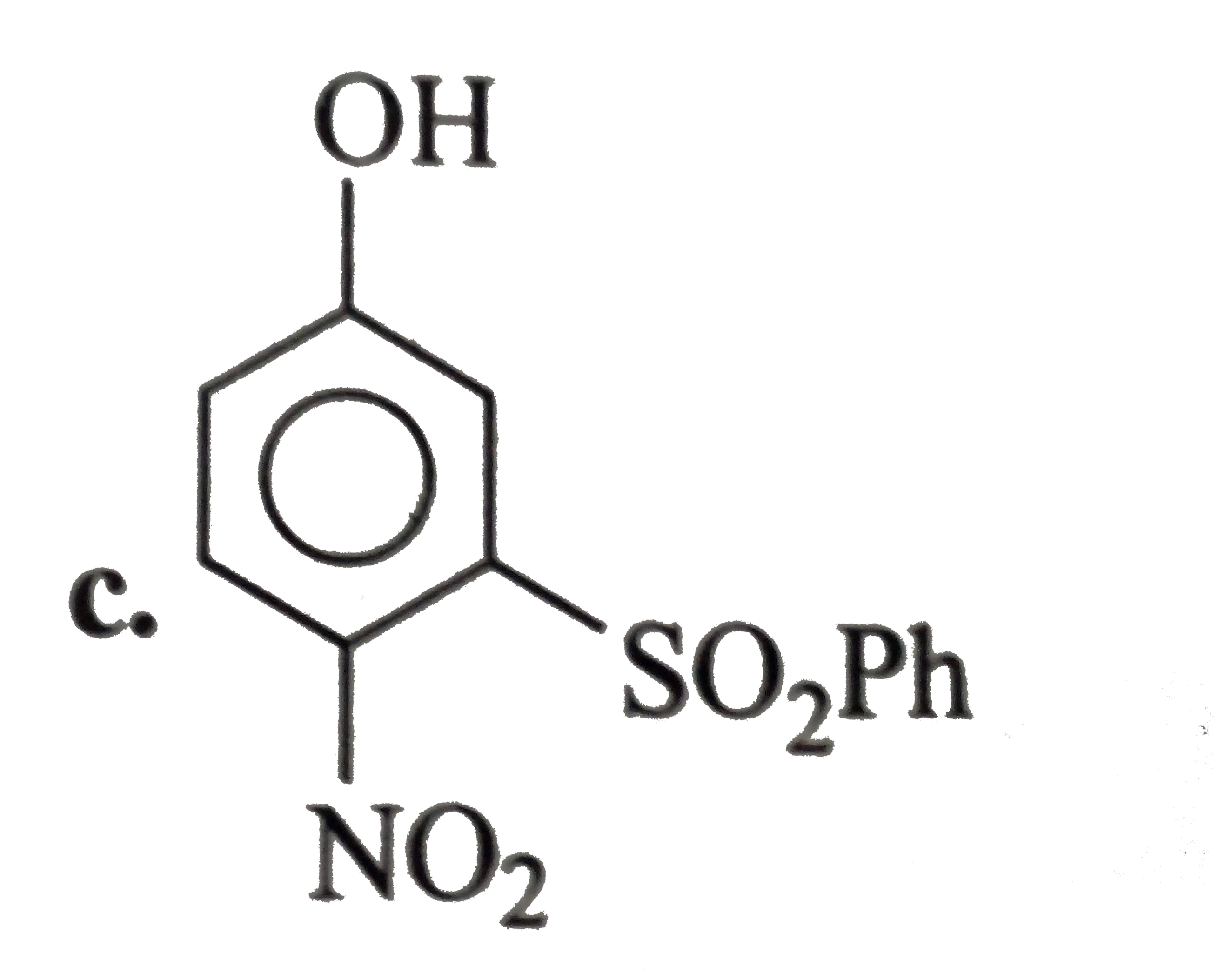
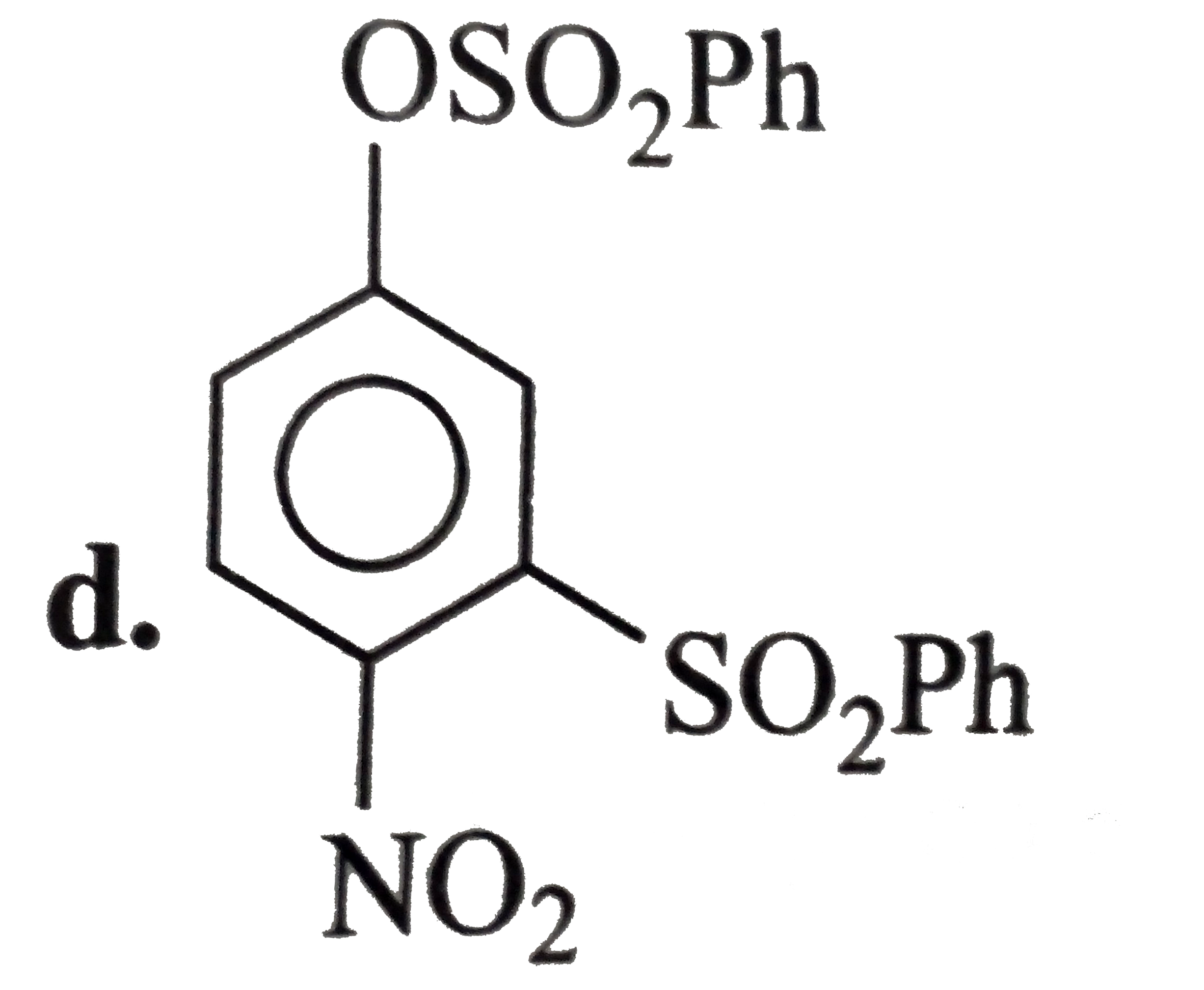
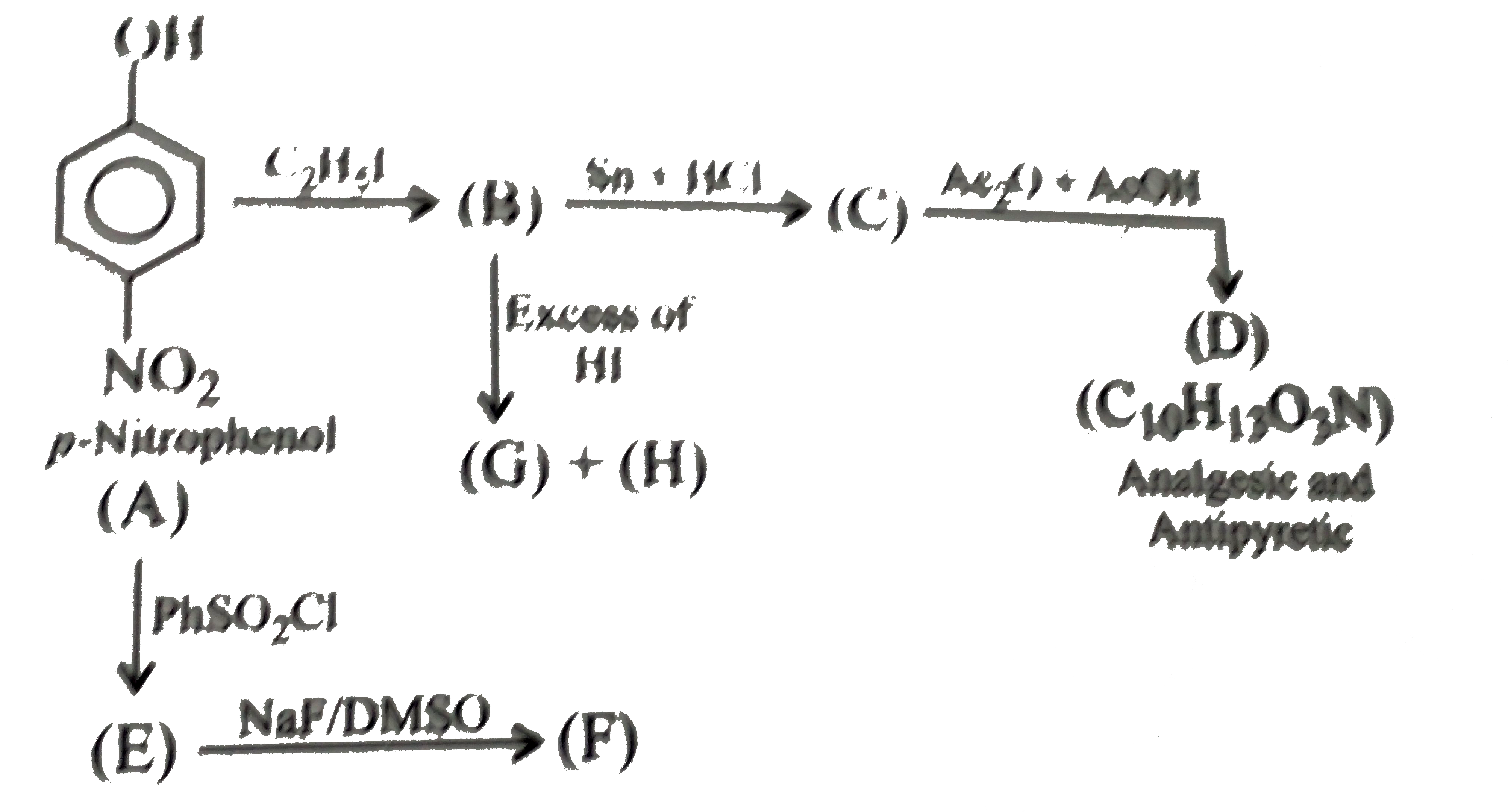A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
ALCOHOL,PHENOL AND ETHERS
CENGAGE CHEMISTRY ENGLISH|Exercise Exercises Multiple Correct|24 VideosALCOHOL,PHENOL AND ETHERS
CENGAGE CHEMISTRY ENGLISH|Exercise Exercises Single Correct|72 VideosALCOHOL,PHENOL AND ETHERS
CENGAGE CHEMISTRY ENGLISH|Exercise Exercises Concept Application|33 VideosALIPHATIC AND AROMATIC ALDEHYDES AND KETONES
CENGAGE CHEMISTRY ENGLISH|Exercise Archives Subjective|26 Videos
Similar Questions
Explore conceptually related problems
CENGAGE CHEMISTRY ENGLISH-ALCOHOL,PHENOL AND ETHERS-Exercises Linked Comprehension
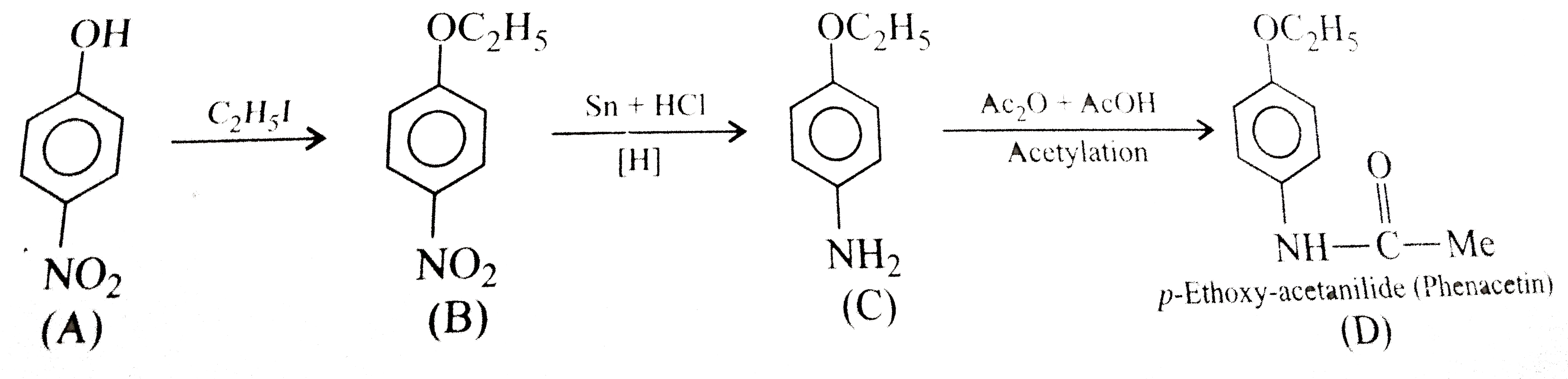
- The compound (D) is:
Text Solution
|
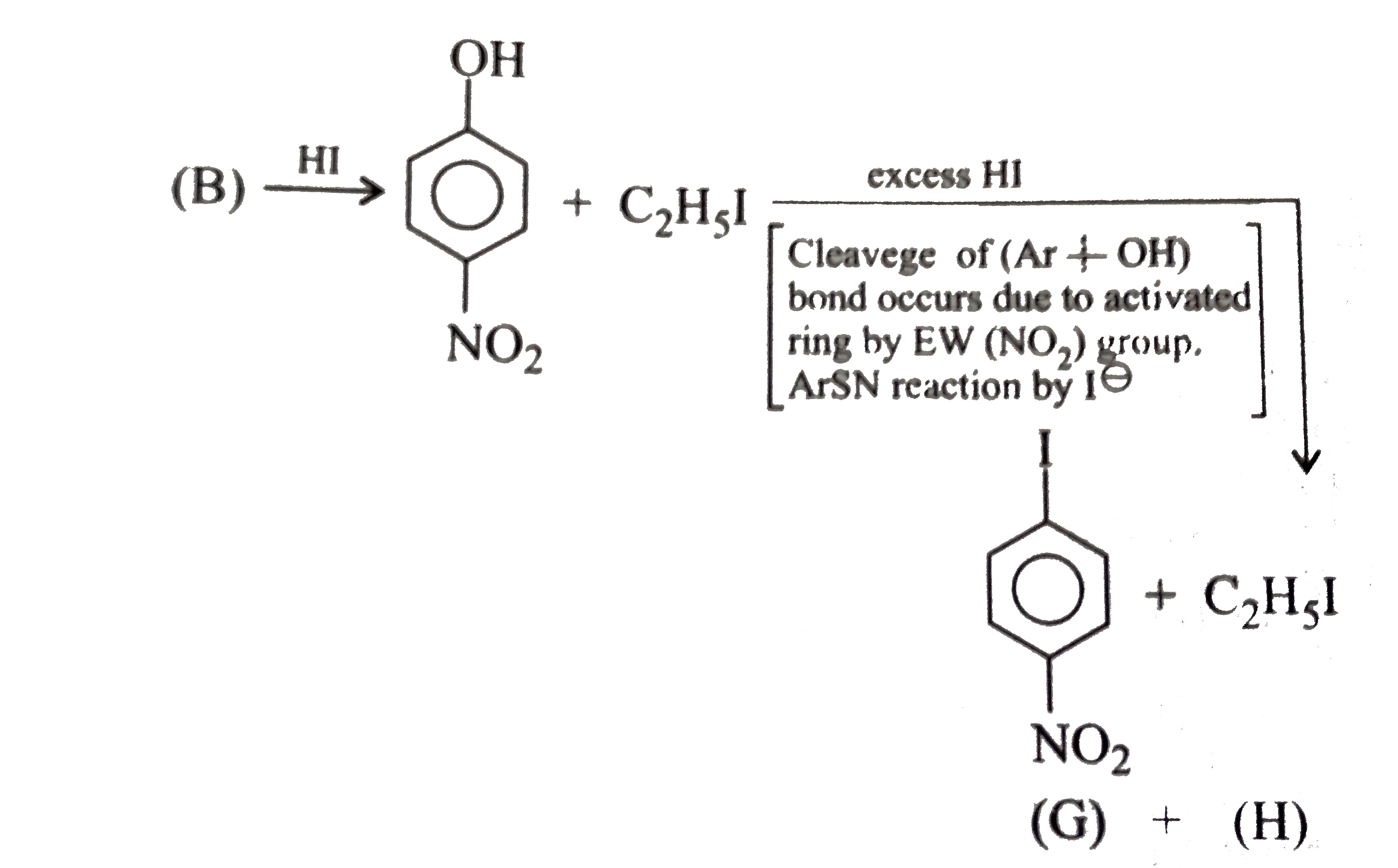
- The compound (G) and (H), respectively, are:
Text Solution
|
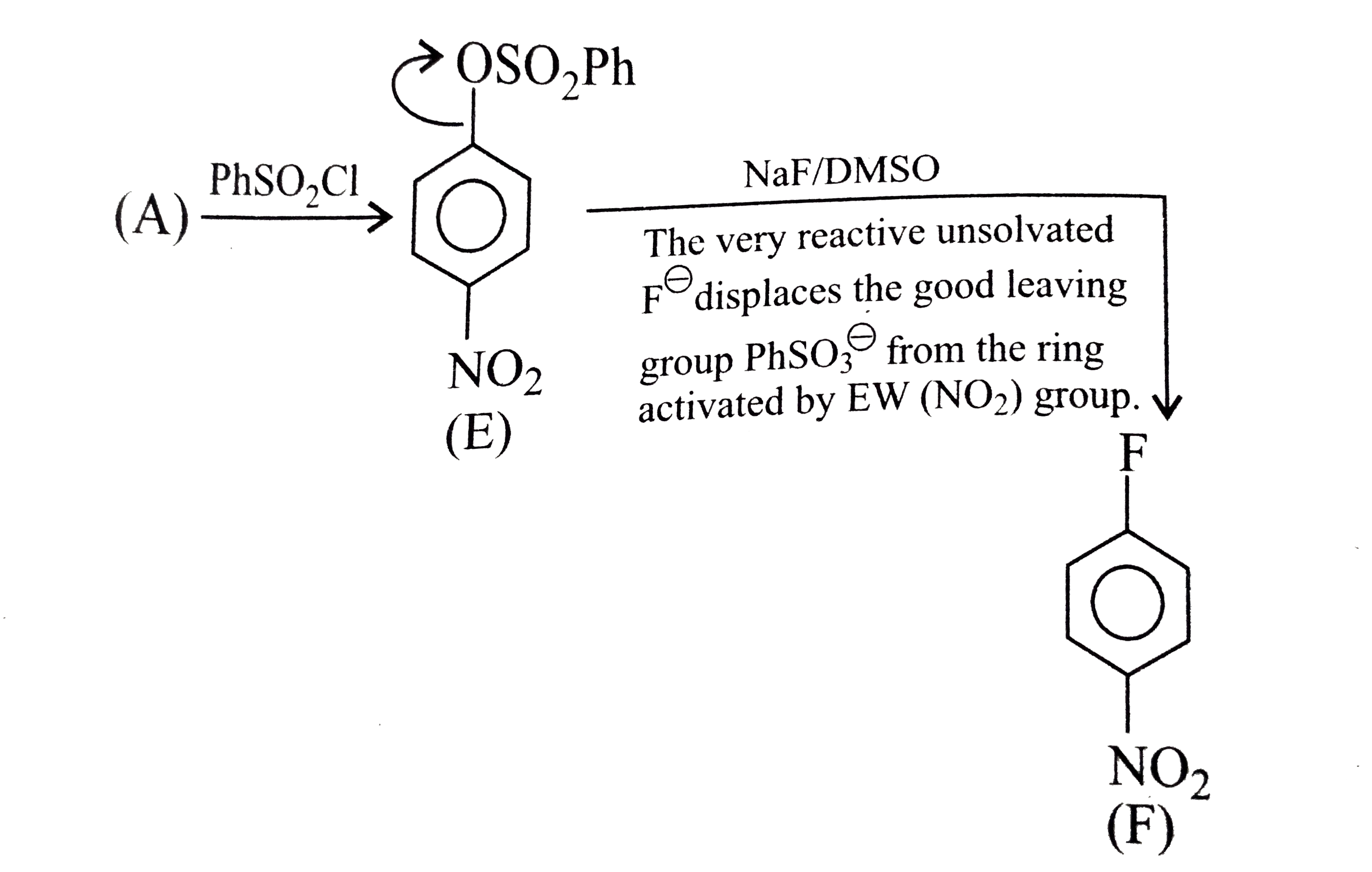
- The compound (E ) is:
Text Solution
|
- The compound (F) is:
Text Solution
|
- The common name of the compound (D) is:
Text Solution
|
- The schotten-Baumann reaction is:
Text Solution
|
- The compound (B) is:
Text Solution
|
- State True or False: Product B is Alcohol.
Text Solution
|
- The compound (B) is:
Text Solution
|
- The major product of the following reaction is: CH(3)C-=CH underset((...
Text Solution
|
- The compound (A) is:
Text Solution
|
- How are the following conversions carried out? Ethyl chloride to pro...
Text Solution
|
- The expected structure of A is?
Text Solution
|
- (a)Give a chemical test to distinguish between saturated and unsaturat...
Text Solution
|
- What physical and chemical properties of elements were used by Mendele...
Text Solution
|
- The compound (C ) is:
Text Solution
|
- The names of reaction and intermediate species involved in the formati...
Text Solution
|
- The compound (D) is:
Text Solution
|
- The compound (F) and (G) are geometrical isomers. The stable form is:
Text Solution
|
- The compound (H) is:
Text Solution
|