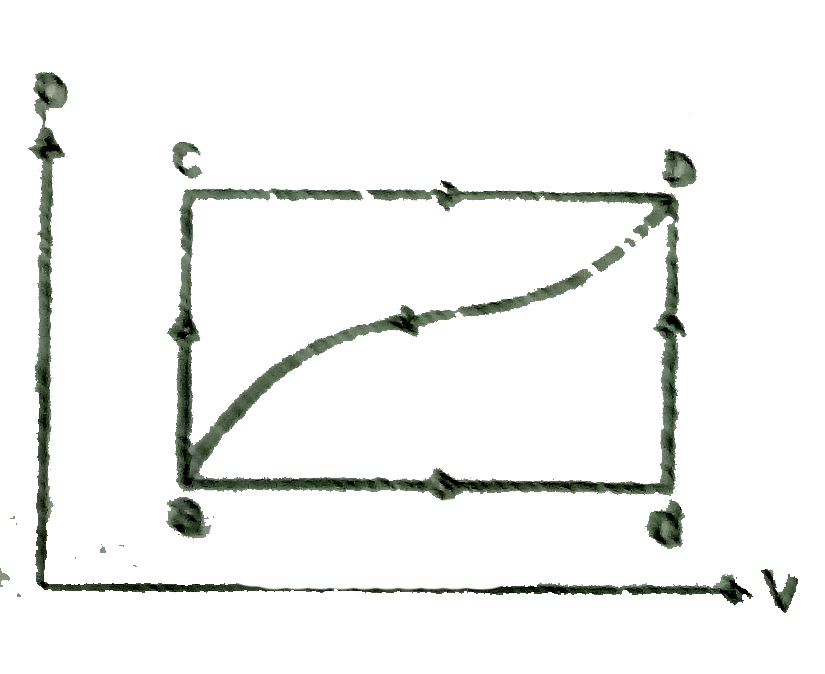
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
KTG & THERMODYNAMICS
RESONANCE ENGLISH|Exercise SECTION (G)|3 VideosKTG & THERMODYNAMICS
RESONANCE ENGLISH|Exercise SECTION (H)|3 VideosKTG & THERMODYNAMICS
RESONANCE ENGLISH|Exercise SECTION (E)|3 VideosKINETIC THEORY OF GASES AND THERMODYNAMICS
RESONANCE ENGLISH|Exercise Exercise|64 VideosMAGNETIC FIELD AND FORCES
RESONANCE ENGLISH|Exercise Exercise|64 Videos
Similar Questions
Explore conceptually related problems