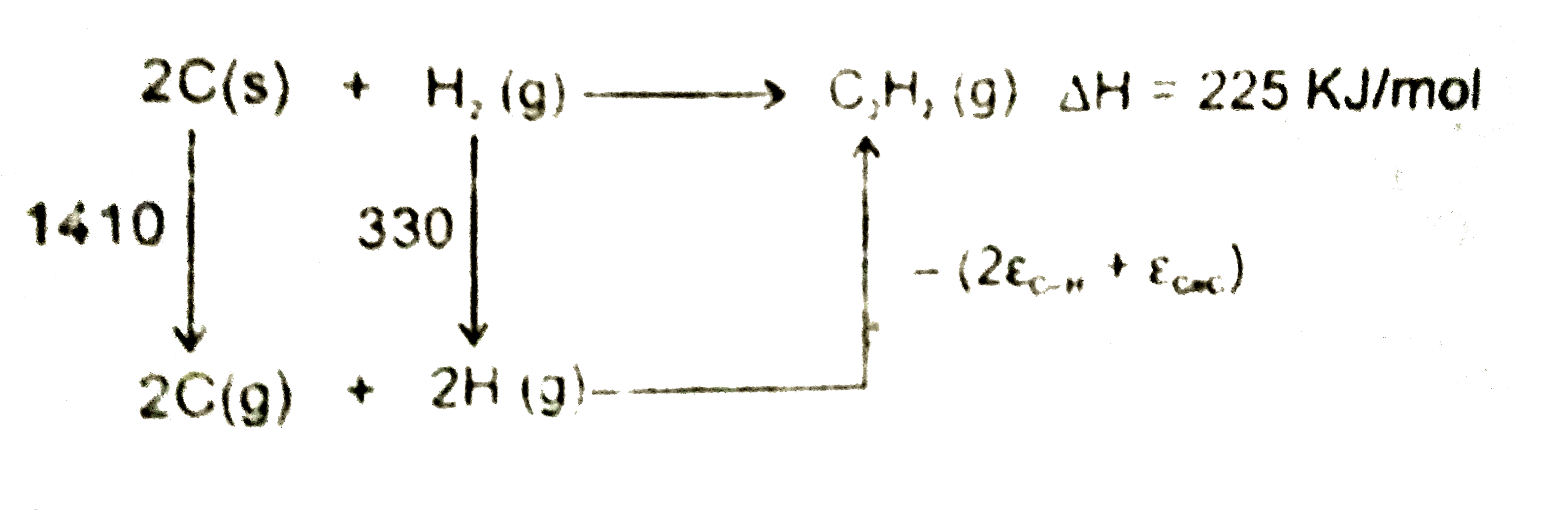A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
THERMODYNAMICS
RESONANCE ENGLISH|Exercise exercise-3 Part-2 :(previous years)|11 VideosTHERMODYNAMICS
RESONANCE ENGLISH|Exercise exercise-3 Part-3 :(Subjective questions)|6 VideosTHERMODYNAMICS
RESONANCE ENGLISH|Exercise exercise-2 Part-4 : Comprehension|5 VideosTEST SERIES
RESONANCE ENGLISH|Exercise CHEMISTRY|50 Videos
Similar Questions
Explore conceptually related problems
RESONANCE ENGLISH-THERMODYNAMICS-exercise-3 Part-1 :(previous years)
- Which of the following equation gives the values of heat of formation ...
Text Solution
|
- In a constant volume calorimeter, 3.5 g of a gas with molecular weight...
Text Solution
|
- The species which by definition has zero standard molar enthalpy of fo...
Text Solution
|
- The bond energy (in kcal mol^(-1)) of a C -C single bond is approximat...
Text Solution
|
- Using the data provided , calculate the multiple bond energy (Kamal^(-...
Text Solution
|
- The standard enthalpies of formation of CO(2)(g), H(2)O(l) and glucos...
Text Solution
|
- When 100mL of 1.0M HCl was mixed with 100 mL of 1.0 M NaOH in an insul...
Text Solution
|
- When 100mL of 1.0M HCl was mixed with 100 mL of 1.0 M NaOH in an insul...
Text Solution
|