Text Solution
Verified by Experts
Topper's Solved these Questions
ELECTROCHEMISRY
RESONANCE ENGLISH|Exercise Miscellaneous Solved Problems|9 VideosELECTROCHEMISRY
RESONANCE ENGLISH|Exercise Board Level Exercise|20 VideosELECTRO CHEMISTRY
RESONANCE ENGLISH|Exercise PHYSICAL CHEMITRY (ELECTROCHEMISTRY)|53 VideosEQUIVALENT CONCEPT & TITRATIONS
RESONANCE ENGLISH|Exercise Part -IV|22 Videos
Similar Questions
Explore conceptually related problems
RESONANCE ENGLISH-ELECTROCHEMISRY-Advanced Level Problems
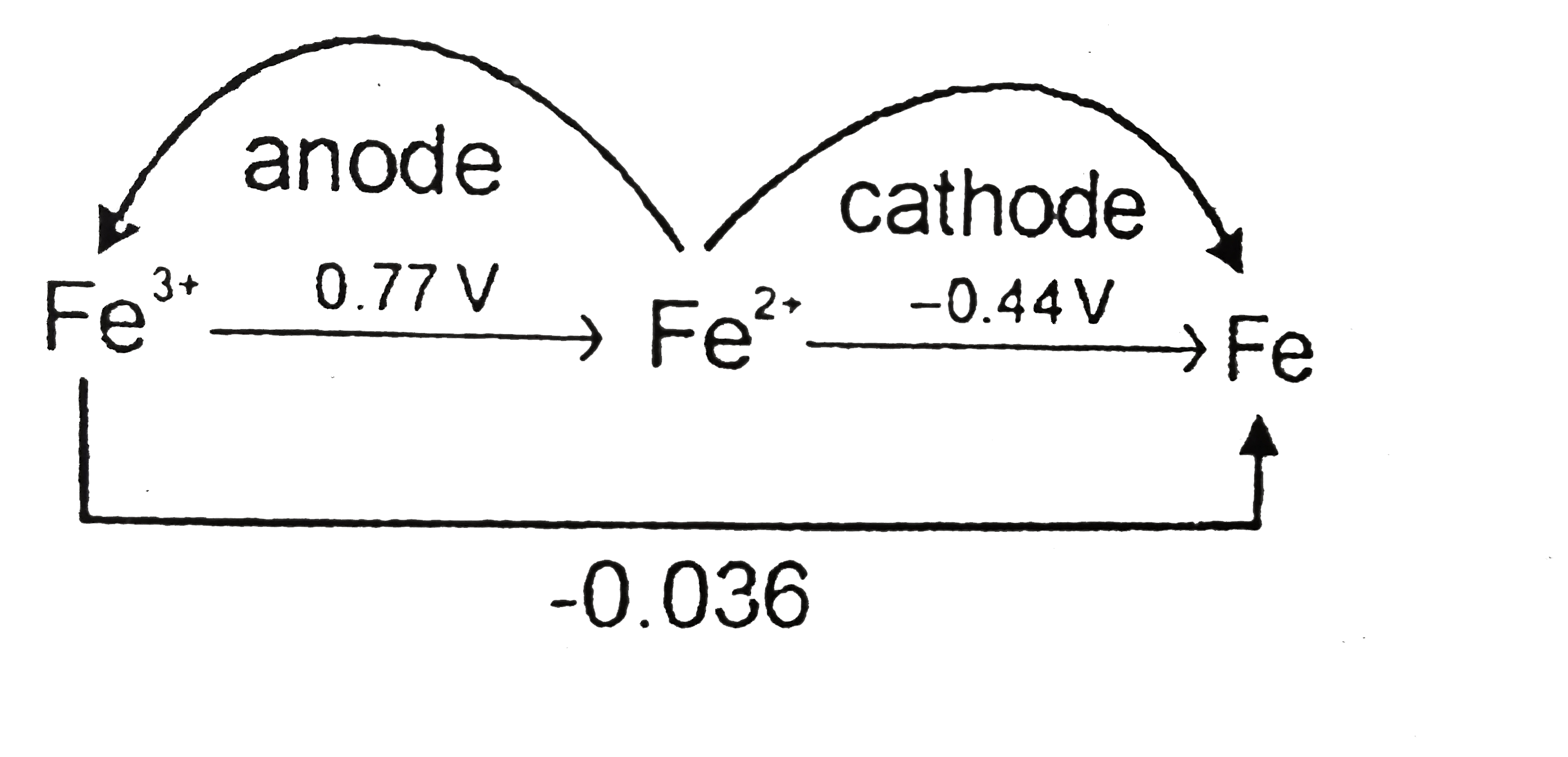
- Will Fe^(2+) disproportionate or not
Text Solution
|
- How much will the reduction potential of a hydrogen electrode change w...
Text Solution
|
- The standard electrode potentials (reduction) of Pt//Fe^(3+),Fe^(+2) a...
Text Solution
|
- An acidic solution of copper (II) sulphate containing some contaiminat...
Text Solution
|
- Acetic acid is titrated with NaOH solution. Which of the following sta...
Text Solution
|
- Which statement is correct?
Text Solution
|
- During the electrolysis of 0.1 M CuSO(4) solution using coppepr electr...
Text Solution
|
- Consider the following Galvanic cell:- By what value the cell vol...
Text Solution
|
- For Pt,Cl(2)(p(1))|HCl(0.1M)|Cl(2)(p(2)), Pt, cell reaction will be sp...
Text Solution
|
- Pt |{:((H(2))),(1atm):}:| pH = 2:|:|:pH =3 |:{:((H(2))Pt),(1atm):}:|. ...
Text Solution
|
- In the given figure the electrolytic cell contains 1L of an aqueous 1M...
Text Solution
|
- By how much will the potential of half cell Cu^(+2)//Cu change if the ...
Text Solution
|
- Which of the following facts is not truegt
Text Solution
|
- Equivalent conductance of 1M CH(3)COOH is 10ohm^(-1)cm^(2) "equIv"^(-1...
Text Solution
|
- Adding powdered Pb and Fe to a solution containing 1.0 M each of Pb^(+...
Text Solution
|
- For the production of X L H2 at STP at cathode, cost of electricity is...
Text Solution
|
- The reaction : Zn(s) + 2AgCl(g) rightarrow ZnCl(2)(aq) + 2Ag(s) oc...
Text Solution
|
- If the pressure of hydrogen gas is increased from 1 atm. To 100 atm, k...
Text Solution
|
- The equilibrium Cu^(+2)(aq)+Cu(s) hArr2Cu^(+) established at 20^(@)C...
Text Solution
|
- The E^(@) in the given figure is
Text Solution
|
- What is cell entropy change of the following cell ? Pt(s)|H(2)(g)|C...
Text Solution
|