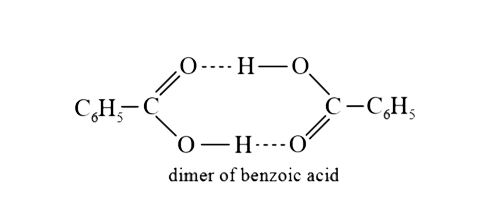
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
LIQUID SOLUTIONS
VMC MODULES ENGLISH|Exercise PRACTICE EXERCISE -01|5 VideosLIQUID SOLUTIONS
VMC MODULES ENGLISH|Exercise PRACTICE EXERCISE -02|6 VideosLIQUID SOLUTIONS
VMC MODULES ENGLISH|Exercise illustrating the concept|5 VideosLIQUID SOLUTION
VMC MODULES ENGLISH|Exercise IMPECCABLE|100 VideosMOCK TEST 1
VMC MODULES ENGLISH|Exercise PART II : CHEMISTRY (SECTION-2)|10 Videos
Similar Questions
Explore conceptually related problems
VMC MODULES ENGLISH-LIQUID SOLUTIONS-SOLVED EXAMPLES
- A weak electrolyte, AB, is 5% dissociated in aqueous solution. What is...
Text Solution
|
- Calculate the boiling point of a 1M aqueous solution (density 1.04 g M...
Text Solution
|
- The vapour pressure of a solvent decreased by 10 mm of Hg when a non-v...
Text Solution
|
- The molal freezing point constant of water is 1.86 K m^(-1). If 342 g...
Text Solution
|
- 20 gm of a binary electrolyte (mol. mass = 100 g/mole) is dissolved in...
Text Solution
|
- Y g of non-volatile or ganic substance of molecular mass M is dissolve...
Text Solution
|
- The values of observed and calculated molecular weights of silver nitr...
Text Solution
|
- At 40^(@)C the vapour pressure in torr,of methyl alcohol - ethyl alcoh...
Text Solution
|
- Calculate the amount of ice that will separate out on cooling containi...
Text Solution
|
- The freezing point of aqueous solution that contains 5% urea by mass, ...
Text Solution
|
- An azeotropic solution of two liquid has boiling point lower than eith...
Text Solution
|
- For a dilute solution, Raoult's low states that :
Text Solution
|
- A molal solution is one that contains 1 mol of a solute in
Text Solution
|
- When mercuric iodide is added to the aqueous solution of KI, then the ...
Text Solution
|
- Which of the following 0.1 M aqueous solutions will have the lowest fr...
Text Solution
|
- The freezing point of equimolal solution will be highest for :
Text Solution
|
- The molecular weight of benzoic acid in benzene as determined by depre...
Text Solution
|
- When 0.004 M Na(2)SO(4) is an isotonic acid with 0.01 M glucose, the d...
Text Solution
|
- The elevation in boiling point of a solution of 13.44 g of CuCl(2) in ...
Text Solution
|
- When 20 g of naphthoic acid (C(11)H(8)O(2)) is dissolved in 50 g of be...
Text Solution
|