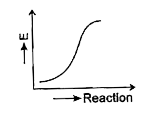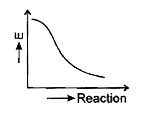A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
CHEMICAL KINETICS
BRILLIANT PUBLICATION|Exercise QUESTION (LEVEL - l) (HOMEWORK) (ASSERTION- REASON) |6 VideosCHEMICAL KINETICS
BRILLIANT PUBLICATION|Exercise QUESTION (LEVEL - II)|36 VideosCARBOXYLIC ACIDS
BRILLIANT PUBLICATION|Exercise LEVEL-II (Assertion -Reason)|4 VideosCO-ORDINATION COMPOUNDS AND ORGANOMETALLICS
BRILLIANT PUBLICATION|Exercise Level-II (Assertion- Reason)|4 Videos
Similar Questions
Explore conceptually related problems
BRILLIANT PUBLICATION-CHEMICAL KINETICS -LEVEL II
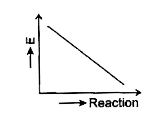
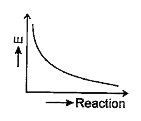
- Which graph shows zero activation energy?
Text Solution
|
- Select the incorrect statement:
Text Solution
|
- The reaction A rarrB+C has rate constant 1xx10^(-3) Ms^(-1). If one s...
Text Solution
|
- For an endothermic reaction where DeltaH represents the enthalpy of re...
Text Solution
|
- In a first order reaction, the concentration of reactant decreases fro...
Text Solution
|
- For the elementary reaction M rarr N, the rate of disappearance of M ...
Text Solution
|
- According to the collision theory of chemical reactions
Text Solution
|
- The activation energy for a simple chemical reaction A rarr B is E(a) ...
Text Solution
|
- The reaction 2NO(g)+O(2)(g)rarr2NO(2)(g) is of first order. Ifvolume ...
Text Solution
|
- For the equilibrium, A(g)rarrB(g), triangleH is - 40 kJ mol^(-1) . If ...
Text Solution
|
- The rate law for a reaction between the substances A and B is given by...
Text Solution
|
- The kinetic data for the given reaction A (g) + 2 B (g) overset(k)rarr...
Text Solution
|
- Reaction A + B rarr C + D follows rate law, r = k[A]^(1//2) [B]^(1//2)...
Text Solution
|
- The reaction A(g) rarr B(g) + 2C(g) is a first order reaction with ra...
Text Solution
|
- For a first order homogenous gaseous reaction A rarr2B +C if the tota...
Text Solution
|
- The gaseous decomposition reaction, A(g) rarr 2B(g) + C(g) is observed...
Text Solution
|
- A gaseous compound A reacts by three independent first order processes...
Text Solution
|
- A compound 'A' dissociate by two parallel first order paths atcertain ...
Text Solution
|
- The reaction cis- X underset(k(b))⇌overset(k(f)) trans-X is first o...
Text Solution
|
- For first order parallel reactions k(1) and k(2) are 4 and 2 min^(- ...
Text Solution
|
- Consider the following first order competing reactions: X overset(k(...
Text Solution
|