A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
THE D- AND F-BLOCK ELEMENT
BRILLIANT PUBLICATION|Exercise LEVEL-II|100 VideosTHE D- AND F-BLOCK ELEMENT
BRILLIANT PUBLICATION|Exercise LEVEL-II (ASSERTION-REASON TYPE)|40 VideosSURFACE CHEMISTRY
BRILLIANT PUBLICATION|Exercise QUESTIONS (LEVEL - II) (ASSERTION REASON TYPE QUESTIONS) |6 VideosTHE D-AND F-BLOCK ELEMENTS
BRILLIANT PUBLICATION|Exercise QUESTIONS (LEVEL-III)|50 Videos
Similar Questions
Explore conceptually related problems
BRILLIANT PUBLICATION-THE D- AND F-BLOCK ELEMENT-LEVEL-II (ASSERTION-REASON TYPE)
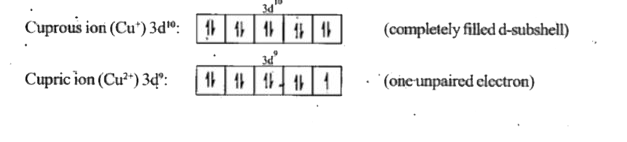
- Cuprous ion is colourless, while cupric ion is coloured because
Text Solution
|
- Assertion : Zn, Cd and Hg are not considered as transition metals. R...
Text Solution
|
- Assertion : Anhydrous copper (II) chloride is covalent while anhydrous...
Text Solution
|
- Assertion : All the members of actinide series are radioactive in natu...
Text Solution
|
- Assertion : Transition metals show variable valency. Reason : Transi...
Text Solution
|
- Assertion : Silver fluoride is soluble in water. Reason : Hydration ...
Text Solution
|
- Assertion : Magnetic moment values of actinides are lesser than the th...
Text Solution
|
- Assertion : MnO is basic whereas Mn2O7, is acidic. Reason : Higher ...
Text Solution
|
- Assertion . The reaction of oxalic acid with acidified KMnO4 proceeds ...
Text Solution
|
- Assertion : In transition elements, radii of 5d series are virtually t...
Text Solution
|
- Assertion : The maximum oxidation state of chromium in its compounds i...
Text Solution
|
- Assertion : Magnetic moment of Mn^(2+) " is less than that of " Cr^(2+...
Text Solution
|
- Assertion : In the series Sc to Zn, the enthalpy of atomisation of zin...
Text Solution
|
- Assertion : In acidic medium, K2 Cr2 O7 " exists as " Cr2 O7^(2-) (ora...
Text Solution
|
- Assertion : Zr and Hf occur together in nature and are difficult to se...
Text Solution
|
- Assertion : The second ionization energies of " "(23) V,""(24)Cr and...
Text Solution
|
- Assertion": Mn'atom loses ns electrons first during ionization as comp...
Text Solution
|
- Assertion : In Cr2O7^(2-) ion, all the Cr-O bond lengths are equal. . ...
Text Solution
|
- Assertion : The purple colour of KMnO4, is due to the charge transfer ...
Text Solution
|
- Assertion : Atomic size of silver is almost the same as that of gold. ...
Text Solution
|
- Assertion : Actinoids show larger number of oxidation states than lant...
Text Solution
|