Text Solution
Verified by Experts
Topper's Solved these Questions
Similar Questions
Explore conceptually related problems
BETTER CHOICE PUBLICATION-SURFACE CHEMISTRY -Question Bank
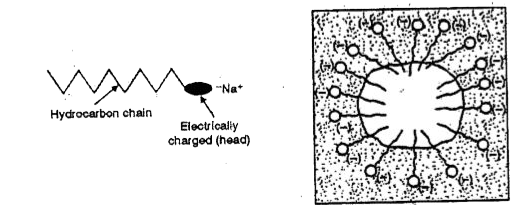
- What are micelles ? Give one example of a micellar system.
Text Solution
|
- Explain the mechanism of the cleansing action of soaps
Text Solution
|
- Discuss cleansing action of soap by micelle formation.
Text Solution
|
- Write short note on peptisation.
Text Solution
|
- What is peptization ? What is cause of peptization ? Give one example.
Text Solution
|
- How will you prepare a collidal solution of gold?
Text Solution
|
- Write short note on electro-dialysis.
Text Solution
|
- Explain Brownian movement.
Text Solution
|
- What name is given to the zig-zag motion of the colloidal particles?
Text Solution
|
- Explain Tyndall effect.
Text Solution
|
- Explain Tyndall effect.
Text Solution
|
- What name is given to the scattering of light by colloidal solution?
Text Solution
|
- Write notes on Hardy Schulze Rule ?
Text Solution
|
- Write short note on Coagulation or flocculation
Text Solution
|
- Write short note on electro-dialysis.
Text Solution
|
- Write short note on Electro-osmosis.
Text Solution
|
- Define electro-osmosis.
Text Solution
|
- What do you understand by protection of colloids ?
Text Solution
|
- What do you mean by gold number ?
Text Solution
|
- What happens when colloidal solutions of Fe(OH)3 and 'AS2O3' are mixe...
Text Solution
|