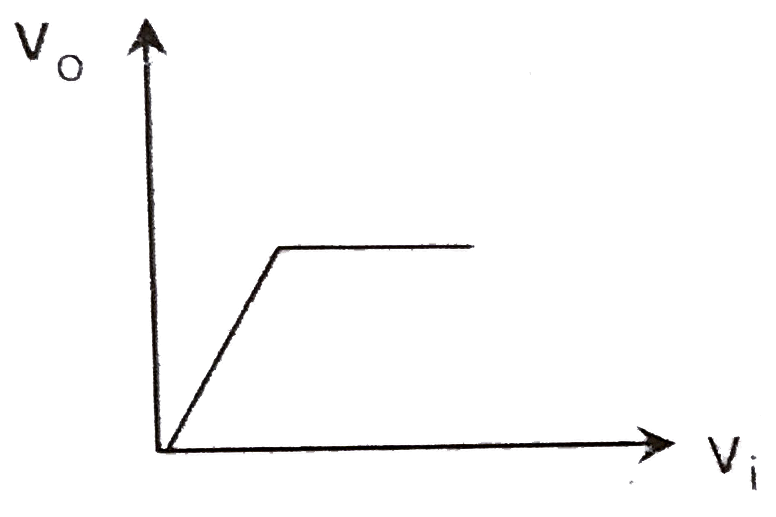
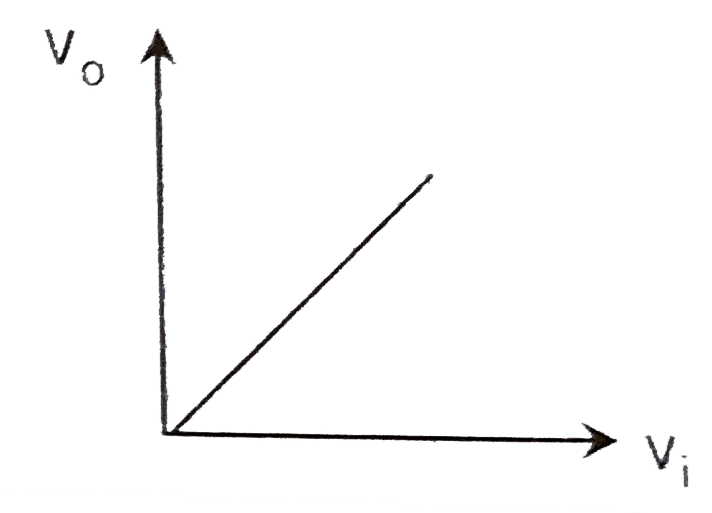
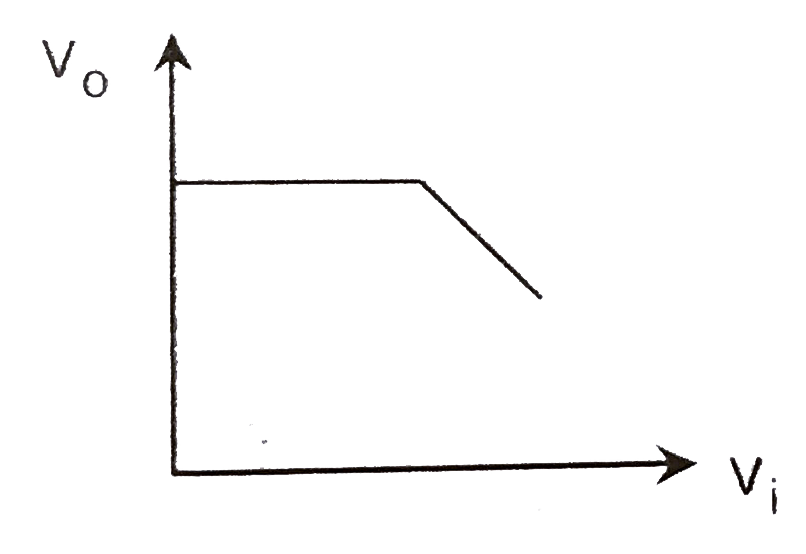
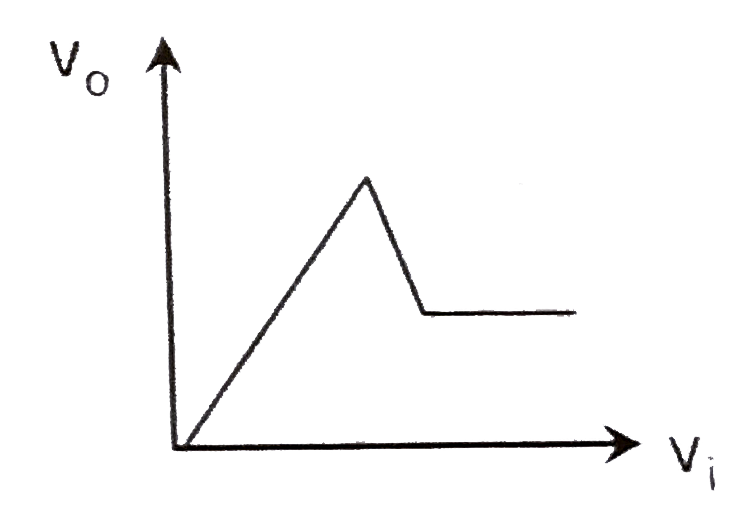
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- Assertion (A): wedge(m(H^(o+))) and wedge(m(overset(c)(O)H)) ions are ...
Text Solution
|
- Assertion: Tr(A)=0 Reason: |A|=1 (A) Both A and R are true and R is th...
Text Solution
|
- Assertion (A): wedge(m(H^(o+))) and wedge(m(overset(c)(O)H)) ions are ...
Text Solution
|
- (a). If both (A) and (R) are correct and (R) is the correct explanatio...
Text Solution
|
- Assertion (A) : In sodium chloride crystal, the coordination number of...
Text Solution
|
- Assertion (A): wedge(m(H^(o+))) and wedge(m(overset(c)(O)H)) ions are ...
Text Solution
|
- Assertion (A): Cu liberates H(2)(g) from a dilute solution of HCl. R...
Text Solution
|
- Assertion (A): Na^(o+) ions are discharged in preference to H^(o+) ion...
Text Solution
|
- Assertion (A) : Cu gets readily corroded in acidic aqueous solution. R...
Text Solution
|