A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
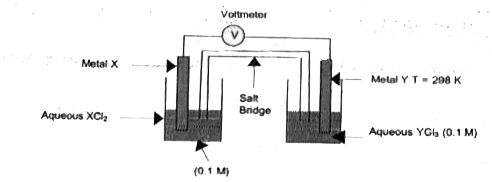
- The following diagram shown as electrochemical cell in which the respe...
Text Solution
|
- The following diagram shows the setup of an electrochemical cell in wh...
Text Solution
|
- Which graph correctly correlates E(cell) as a function of concentratio...
Text Solution
|
- Calculate the standard cell potentials of the galvanic cells in which ...
Text Solution
|
- The logarithm of the equilibriium constant of the cell reaction cor...
Text Solution
|
- The following diagram shown as electrochemical cell in which the respe...
Text Solution
|
- The following diagram shown as electrochemical cell in which the respe...
Text Solution
|
- The following diagram shown as electrochemical cell in which the respe...
Text Solution
|
- The following diagram shown as electrochemical cell in which the respe...
Text Solution
|