Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
Similar Questions
Explore conceptually related problems
HC VERMA-LAWS OF THERMODYNAMICS-Exercises
- A 100 kg block is started with a speed of 2.0 m s^(-1) on a long, roug...
Text Solution
|
- calculate the change in internal energy of a gas kept in a rigid conta...
Text Solution
|
- the pressure of a gas change linearly with volume from 10kPa, 200 cc t...
Text Solution
|
- An ideal gas is taken from an initial state i to a final state f in s...
Text Solution
|
- shows three paths through which a gas can be taken from the state A to...
Text Solution
|
- when a system is taken through the process abc shown in the fig. 80 J ...
Text Solution
|
- 50 cal of heat should be supplied to take a system from the state A t...
Text Solution
|
- calculate the heat absorbed by a system in going through the cyclic pr...
Text Solution
|
- In the given figure, the value of x is
Text Solution
|
- A substance is taken through the process abc as shown in fig, if the ...
Text Solution
|
- A gas is taken along the path AB as shown in fig, if 70 cal of heat is...
Text Solution
|
- The internal energy of a gas is given by U=1.5pV. It expands from 100...
Text Solution
|
- A gas is enclosed in a cylindrical vessel fitted with a frictionless p...
Text Solution
|
- A gas is initially at a pressure of 100 kPa and its volume is 2.0 m^(3...
Text Solution
|
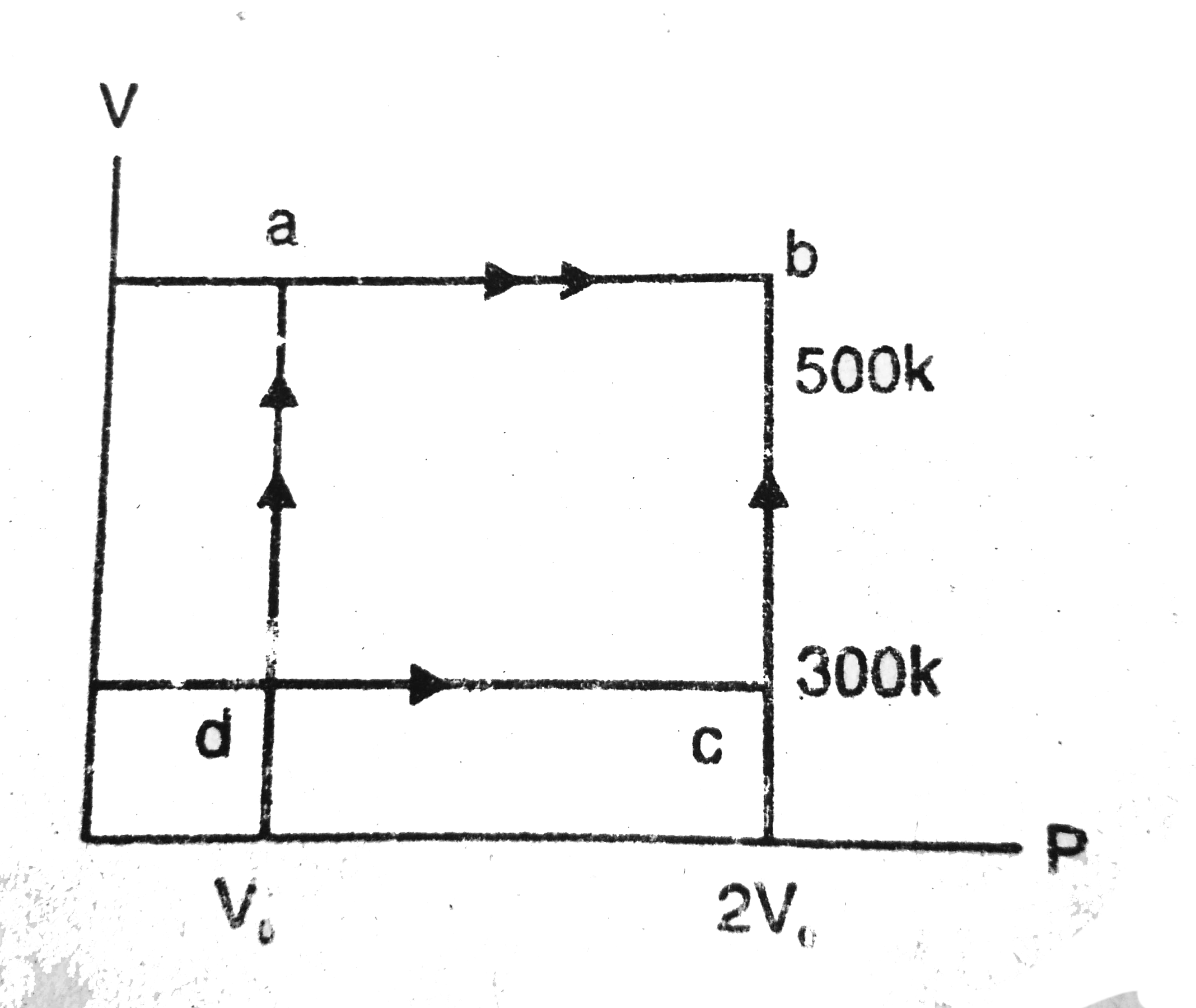
- Consider the cyclic process ABCA, shown in fig, performed on a sample ...
Text Solution
|
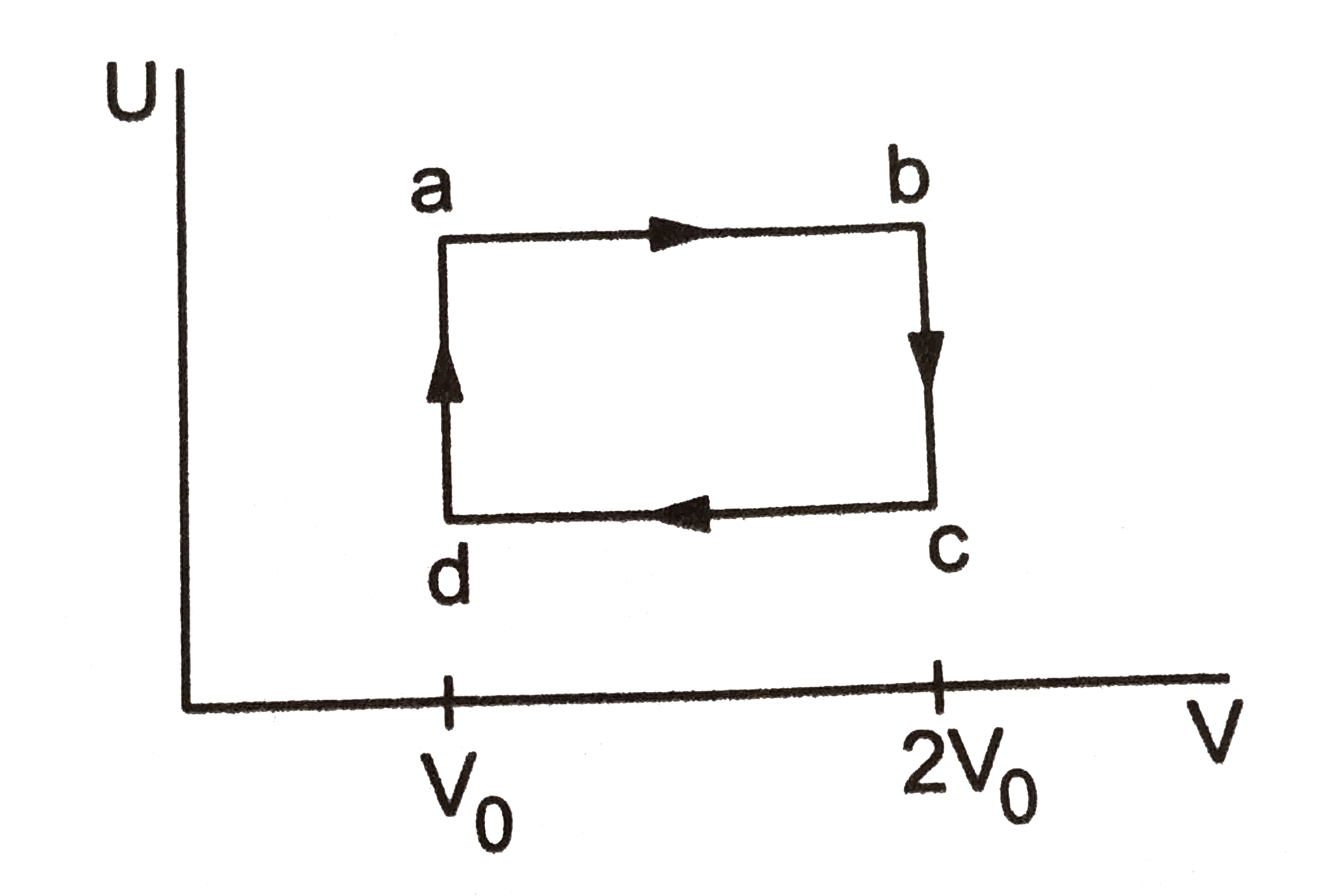
- Fig shows the variation in the internal energy U with the volume V of ...
Text Solution
|
- Find the change in the internal energy of 2 kg of water as it heated f...
Text Solution
|
- Calculate the increase in the internal energy of 10 g of water when it...
Text Solution
|
- Fig shows a cylindrical tube of volume V with adiabatic walls containi...
Text Solution
|
- An adiabatic vessel of total volume V is divided into two equal parts ...
Text Solution
|