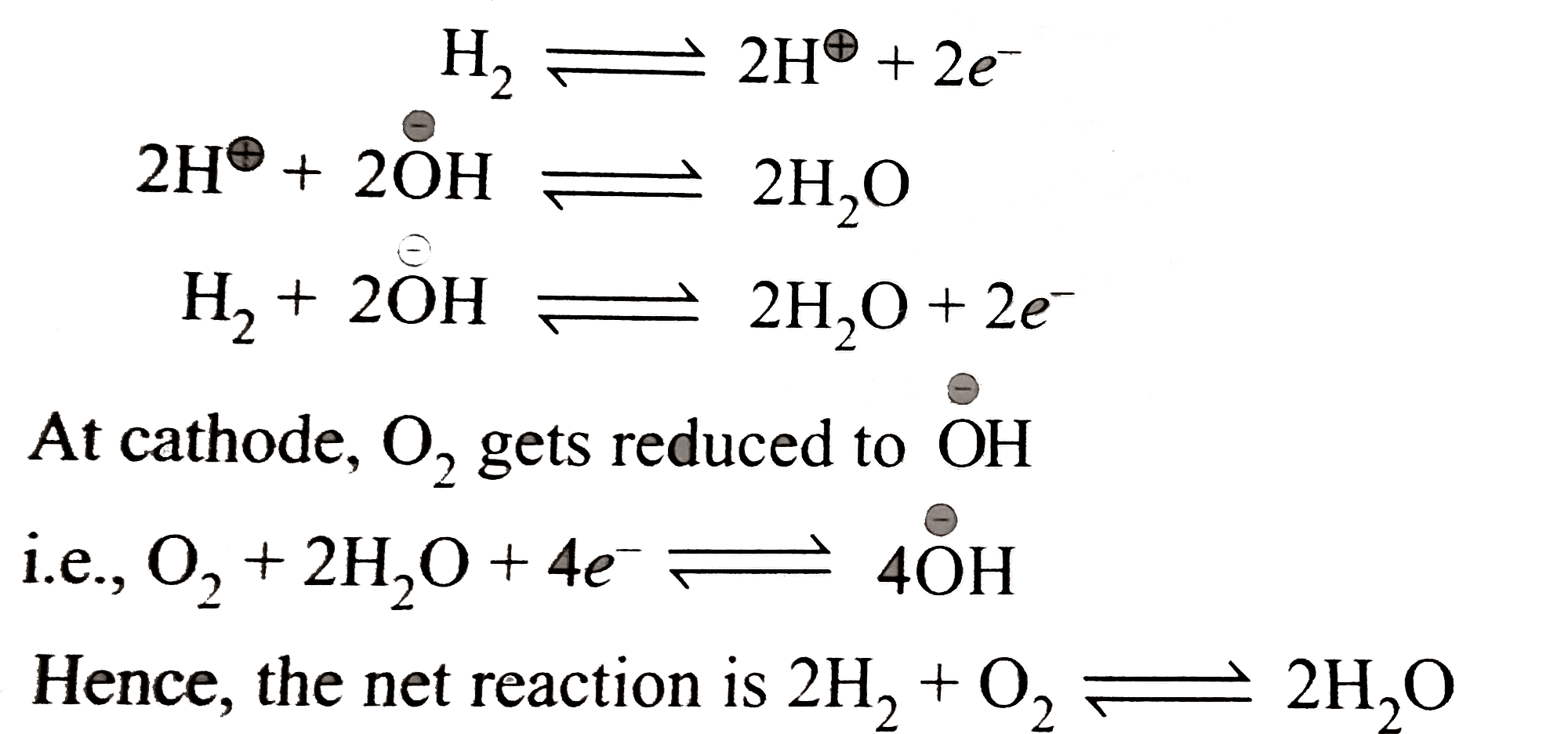A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- Fuel cells : Fuel cells are galvanic cells in which the chemical energ...
Text Solution
|
- Fuel cells : Fuel cells are galvanic cells in which the chemical energ...
Text Solution
|
- Fuel cells : Fuel cells are galvanic cells in which the chemical energ...
Text Solution
|
- Fuel cells : Fuel cells are galvanic cells in which the chemical energ...
Text Solution
|
- Fuel cells : Fuel cells are galvanic cells in which the chemical energ...
Text Solution
|
- ईधन सेल क्या हैं। हाइड्रोजन –ऑक्सीजन ईधन सेल का वर्णन संक्षेप में कीज...
Text Solution
|
- ईंधन सेल में ईंधन ऊर्जा को विद्युत ऊर्जा में बदला जाता है।
Text Solution
|
- What are fuel cells ? Discuss briefly hydrogen-oxygen fuel cell ?
Text Solution
|
- What are fuel cells ? Write the electrode reactions of a fuel cell whi...
Text Solution
|