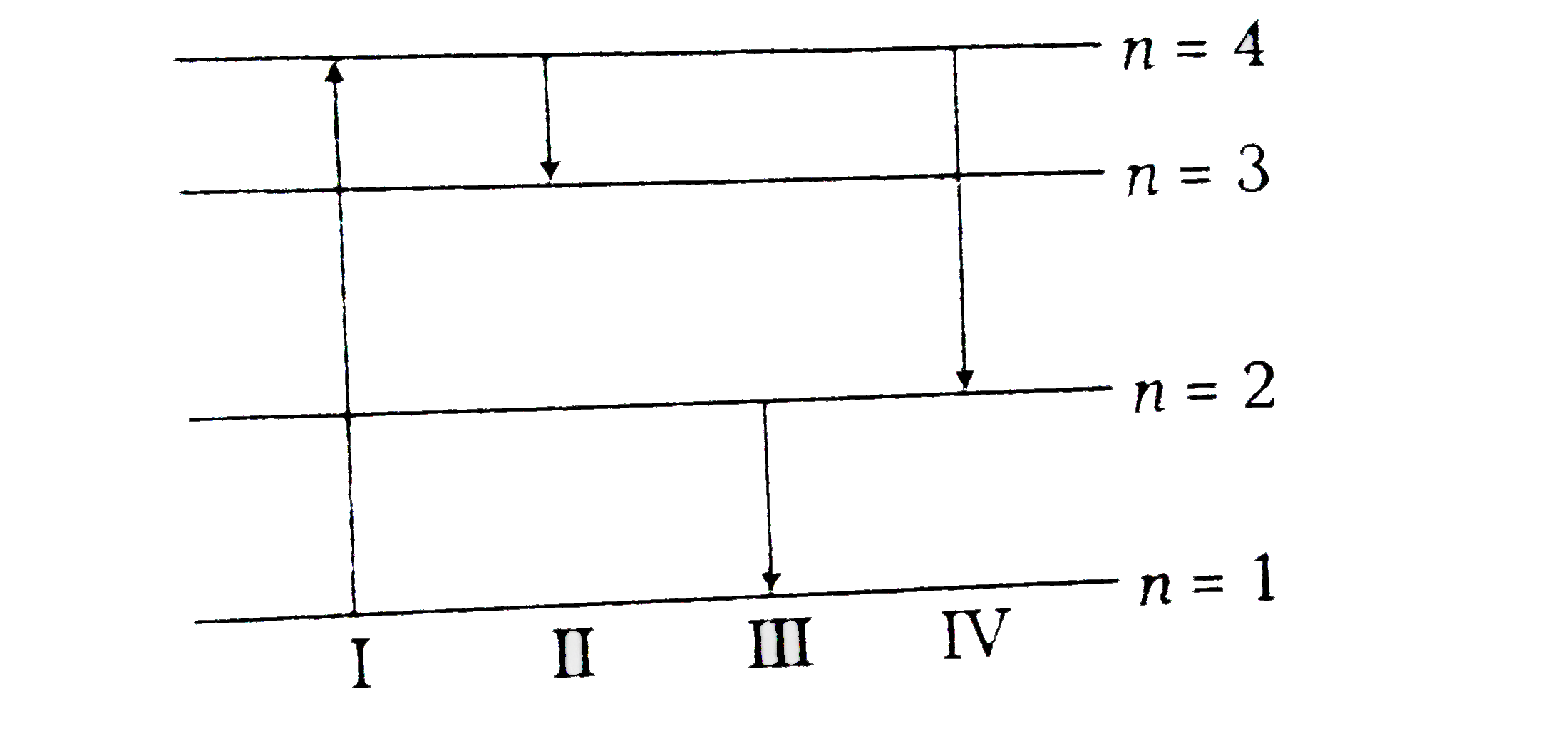A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
ATOMS
AAKASH SERIES|Exercise EXERCISE - II|19 VideosATOMS
AAKASH SERIES|Exercise PRACTICE EXERCISE|21 VideosATOMS
AAKASH SERIES|Exercise PROBLEM|24 VideosAPPENDICES (REVISION EXERCISE)
AAKASH SERIES|Exercise LAW OF MOTION|128 VideosCAPACITORS
AAKASH SERIES|Exercise PRACTICE SHEET (ADVANCED) (Integer Type Questions)|2 Videos
Similar Questions
Explore conceptually related problems
AAKASH SERIES-ATOMS-EXERCISE - I
- According to Bohr's principle , the relation between principle quantum...
Text Solution
|
- The diagram shows the energy levels for an electron in a certain atom....
Text Solution
|
- The wavelengths involved in the spectrum of deuterium (.(1)^(2)D) are...
Text Solution
|
- Hydrogen atom does not emit X-rays because
Text Solution
|
- Atomic hydrogen is excited from the grounds state to the n^(th) state....
Text Solution
|
- Which of the following is not correct about bohr model of the hydrogen...
Text Solution
|
- Which of the following transitions gives photon of maximum energy
Text Solution
|
- The Bohr model of atoms
Text Solution
|
- Bohr's model of hydrogen atom.
Text Solution
|
- Three photons coming from excited atomic-hydrogen sample are picked up...
Text Solution
|
- Whenever a hydrogen atom emitsa photon in the Balmer series .
Text Solution
|
- When a hydrogen atom is raised from the ground state to an excited sta...
Text Solution
|
- Consider a spectral line resulting from the transition n = 5 to n = 1,...
Text Solution
|
- In which of the following systems will be the radius of the first orbi...
Text Solution
|
- If the radius of an orbit is r and the velocity of electron in it is v...
Text Solution
|
- As the n (number of orbit) increases, the difference of energy between...
Text Solution
|
- Which of the following parameters are the same for all hydrogen like a...
Text Solution
|
- The electron in a hydrogen atom makes a transition from an excited sta...
Text Solution
|
- The angular momentum of an electron in a hydrogen atom is proportional...
Text Solution
|
- The equation corresponding to the wave number of spectral lines in Pfu...
Text Solution
|