A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
THERMODYNAMICS
ERRORLESS|Exercise NCERT BASED QUESTIONS (ISOTHERMAL PROCESS)|16 VideosTHERMODYNAMICS
ERRORLESS|Exercise NCERT BASED QUESTIONS (ADIABATIC PROCESS)|31 VideosSURFACE TENSION
ERRORLESS|Exercise ASSERTION & REASON|11 VideosTHERMOMETRY, THERMAL EXPANSION AND CALORIMETRY
ERRORLESS|Exercise ASSERTION & REASON|10 Videos
Similar Questions
Explore conceptually related problems
ERRORLESS-THERMODYNAMICS-ASSERTION AND REASON
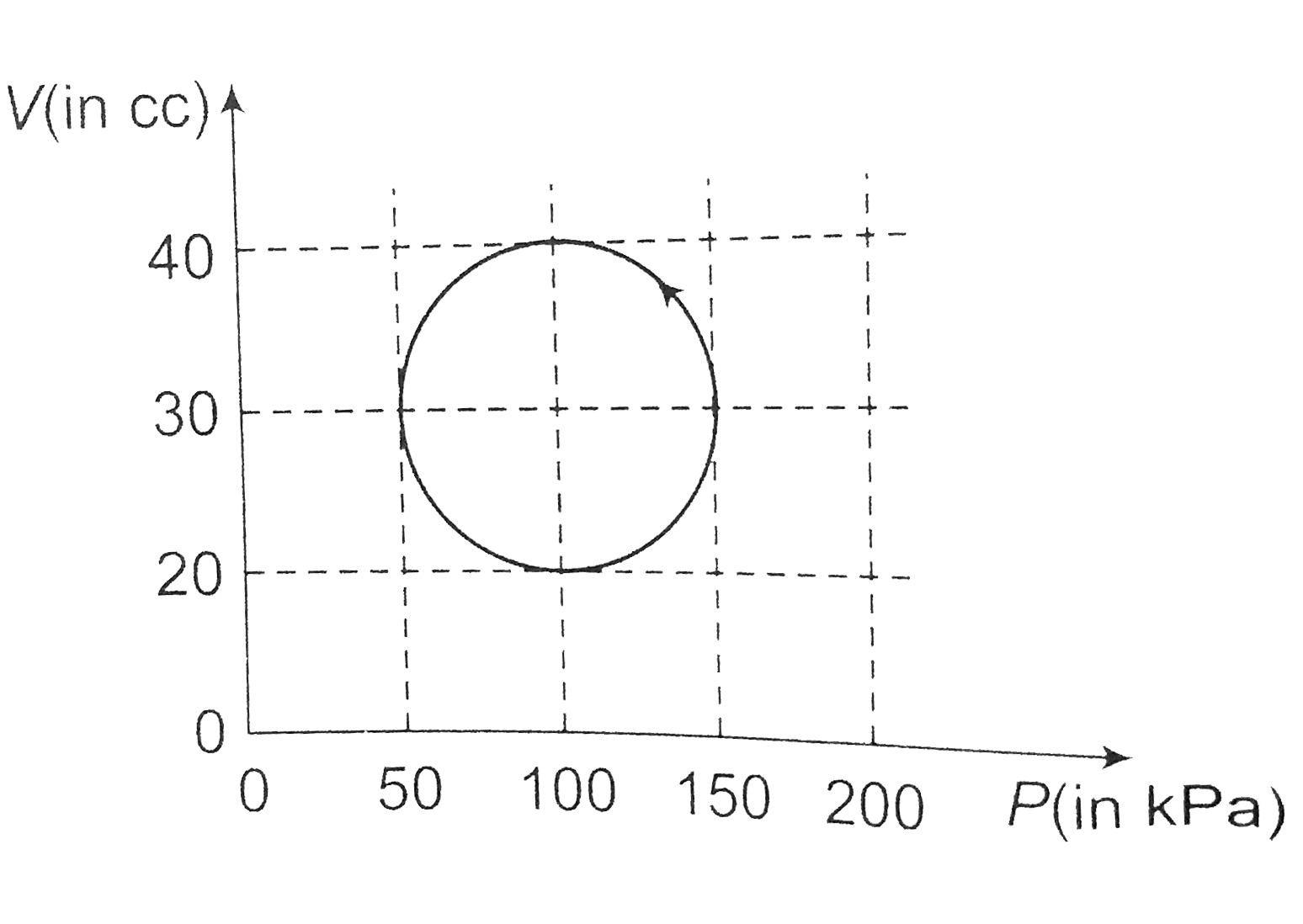
- A system is taken through a cyclic process represented by a circle as ...
Text Solution
|
- Assertion: Reversible systems are difficult to find in real world. R...
Text Solution
|
- Assertion: Air quickly leaking out of a balloon becomes coolers. Rea...
Text Solution
|
- Aseertion: Thermodynamics process in nature are irreversible. Reason...
Text Solution
|
- Assertion: When a bottle of cold carbonated drink is opened, a slight...
Text Solution
|
- Assertion: The isothermal curves intersect each other at a certain poi...
Text Solution
|
- Assertion: In adiabatic compression, the internal energy and temperatu...
Text Solution
|
- Statement-1: In isothermal process whole of the heat energy suppled to...
Text Solution
|
- Assertion : We can not change the temperature of a body without givin...
Text Solution
|
- Statement I: The specific heat of a gas in an adiabatic process is zw...
Text Solution
|
- Assertion: The isothermal curves intersect each other at a certain poi...
Text Solution
|
- Assertion: The heat supplied to a system is always equal to the increa...
Text Solution
|
- Assertion: The temperature of the surface of the sun is approximately ...
Text Solution
|
- Assertion : It is not possible for a system, unaided by an external a...
Text Solution
|
- Assertion : If an electric fan be switched on in a closed room, the a...
Text Solution
|
- Assertion : When a glass of hot milk is placed in a room and allowed t...
Text Solution
|
- Statement-1 : In an adiabatic process, change in internal energy of a ...
Text Solution
|
- Assertion : The Carnot cycle is useful in understanding the performanc...
Text Solution
|
- A : Work done by a gas in isothermal expension is more than the work d...
Text Solution
|
- Assertion : First law of thermodynamics is a restatement of the princi...
Text Solution
|
- Assertion : Zeroth law of thermodynamic explain the concept of energy...
Text Solution
|