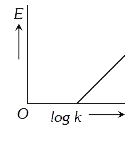
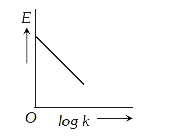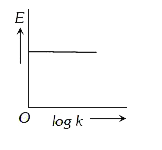A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- EMF of a cell is related to standard EMF,E^(@) by the equation E=E^(...
Text Solution
|
- If the DeltaG^(@) of a cell reaction AgCI+e^(-) is -21.20KJ stand...
Text Solution
|
- If n cells each of emf E and internal resistance are connected in para...
Text Solution
|
- Assertion: The emf of a cell is related with equilibrium constant of t...
Text Solution
|
- If the DeltaG of a cell reaction AgCl+e^(-)toAg+Cl^(-) is -21.20 kJ, t...
Text Solution
|
- n electric cells having emf e and internal resistance r each are conne...
Text Solution
|
- n electric cells of emf e and internal resistance r each are connected...
Text Solution
|
- If standard emf (E^(@)) of a half -cell Y^(2+)//Y^(+) is 0.15 V, the s...
Text Solution
|
- The standard reduction potentials E^(@) for the half reactions are as ...
Text Solution
|