Similar Questions
Explore conceptually related problems
Recommended Questions
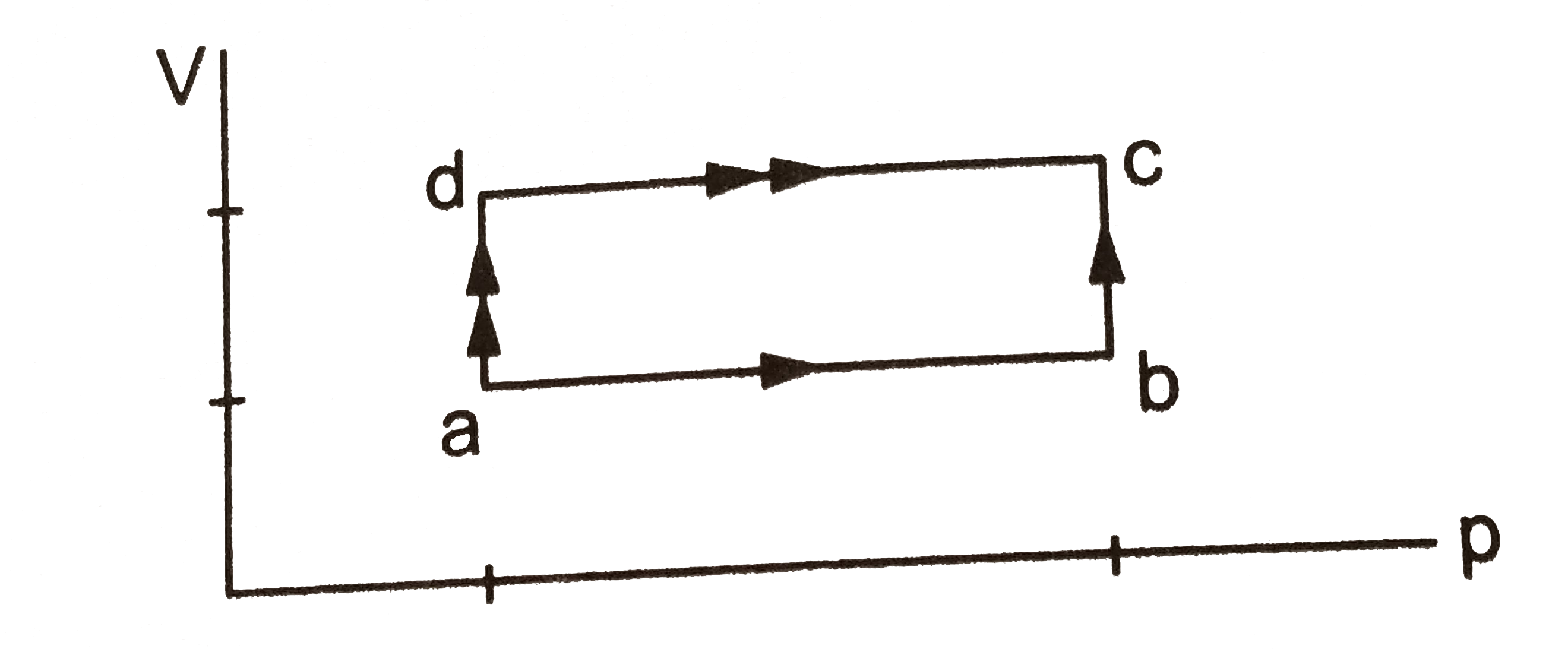
- when a system is taken through the process abc shown in 80 J of heat i...
Text Solution
|
- when a system is taken through the process abc shown in 80 J of heat i...
Text Solution
|
- When a system is taken through the process abc shown in figure, 80 J o...
Text Solution
|
- A system is provided 50 J of heat and work can be done on the system i...
Text Solution
|
- In a process, 701 J of heat is absorbed by a system and 394 J of work ...
Text Solution
|
- When a system is taken from A to C through path ABC, 10 J of heat flow...
Text Solution
|
- The work done by a system is 8 J, when 40 J heat is supplied to it. Th...
Text Solution
|
- In a process 701 J of heat is absorbed by a system and 394 J of work i...
Text Solution
|
- In a process, 701 J of heat is absorbed by a system and 394 J of work ...
Text Solution
|