Similar Questions
Explore conceptually related problems
Recommended Questions
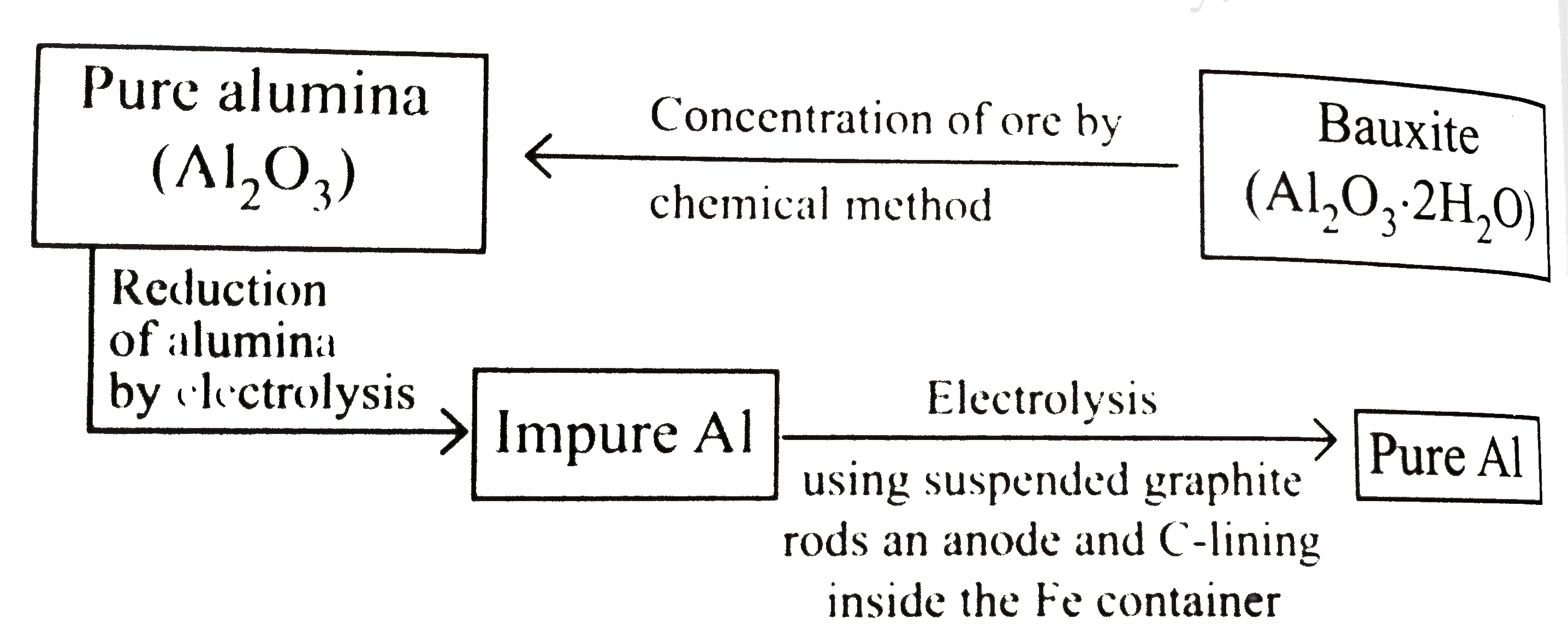
- Extraction of aluminium can be understood by : Electrolytric redu...
Text Solution
|
- Extraction of aluminium can be understood by : Electrolytric redu...
Text Solution
|
- Extraction of aluminium can be understood by : Electrolytric redu...
Text Solution
|
- Extraction of aluminium can be understood by : Electrolytric reduction...
Text Solution
|
- Extraction of aluminium can be understood by : Electrolytric reduction...
Text Solution
|
- Electrolytic reduction of Al(2)O(3) . Electrolyte ( Al(2)O(3)+"cryolit...
Text Solution
|
- Extraction of aluminium can be understood by : Electrolytric redu...
Text Solution
|
- Extraction of aluminium can be understood by : Electrolytric redu...
Text Solution
|
- Extraction of aluminium can be understood by : Electrolytric redu...
Text Solution
|