Text Solution
Verified by Experts
Topper's Solved these Questions
P-BLOCK ELEMENTS
VIKRAM PUBLICATION ( ANDHRA PUBLICATION)|Exercise GROUP 18 ELEMENTS (LONG ANSWER QUESTIONS)|1 VideosORGANIC COMPOUNDS CONTAINING NITROGEN
VIKRAM PUBLICATION ( ANDHRA PUBLICATION)|Exercise DAM SURE (SAQ - 8 Marks)|15 VideosPOLYMERS
VIKRAM PUBLICATION ( ANDHRA PUBLICATION)|Exercise INTEXT QUESTIONS|6 Videos
Similar Questions
Explore conceptually related problems
VIKRAM PUBLICATION ( ANDHRA PUBLICATION)-P-BLOCK ELEMENTS -INTEXT QUESTIONS
- List the important sources of sulphur.
Text Solution
|
- Write the order of thermal stability of the hydrides of group 16 eleme...
Text Solution
|
- Why is H(2)O a liquid and H(2)S a gas ?
Text Solution
|
- Which of the following does not react with oxygen directly ? Zn, Ti, P...
Text Solution
|
- Complete the following reactions. i) C(2)H(4)+O(2)to ii) 4" Al"+3O(2...
Text Solution
|
- Why does O(3) act as a powerful oxidising agent ?
Text Solution
|
- How is O(3) estimated quantitatively ?
Text Solution
|
- What happens when sulphur dioxide is passed through an aqueous solutio...
Text Solution
|
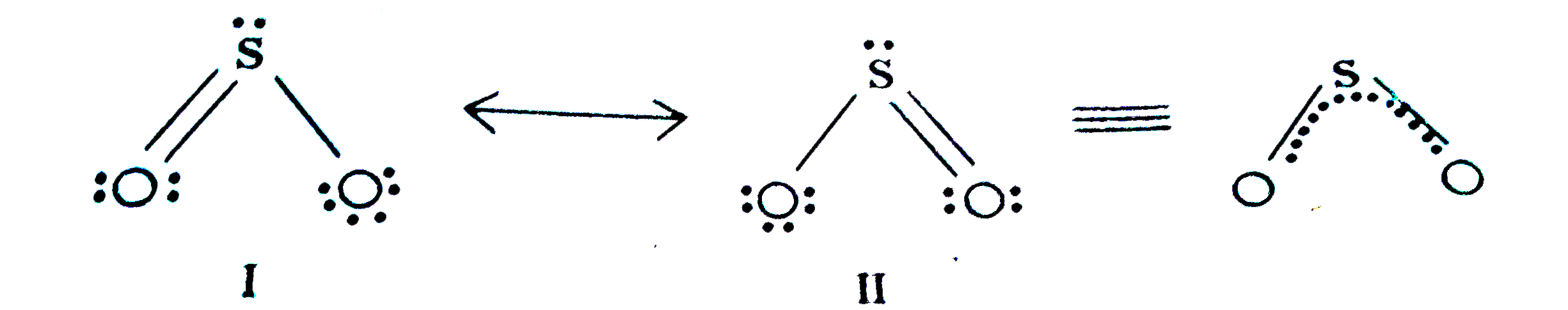
- Comment on the nature of two S - O bonds formed is SO(2) molecule. Are...
Text Solution
|
- How is the presence of SO(2) detected ?
Text Solution
|
- Mention three areas in which H(2)SO(4) plays an important role.
Text Solution
|
- Write the conditions to maximize the yield of H(2)SO(4) by contact pro...
Text Solution
|
- Considering the parameters such as bond dissociation enthalpy, electro...
Text Solution
|
- Give two examples to show the anomalous behaviour of flurine.
Text Solution
|
- Sea is the greatest source of some halogens. Comment.
Text Solution
|
- Give the reason for bleaching action of Cl(2) ?
Text Solution
|
- Name some poisonous gases which can be prepared from chlorine gas.
Text Solution
|
- Why is helium used in diving apparatus ?
Text Solution
|
- Balance the following equation : XeF(6)+H(2)OtoXeO(2)F(2)+HF
Text Solution
|
- Why has it been difficult to study the chemistry of radon ?
Text Solution
|