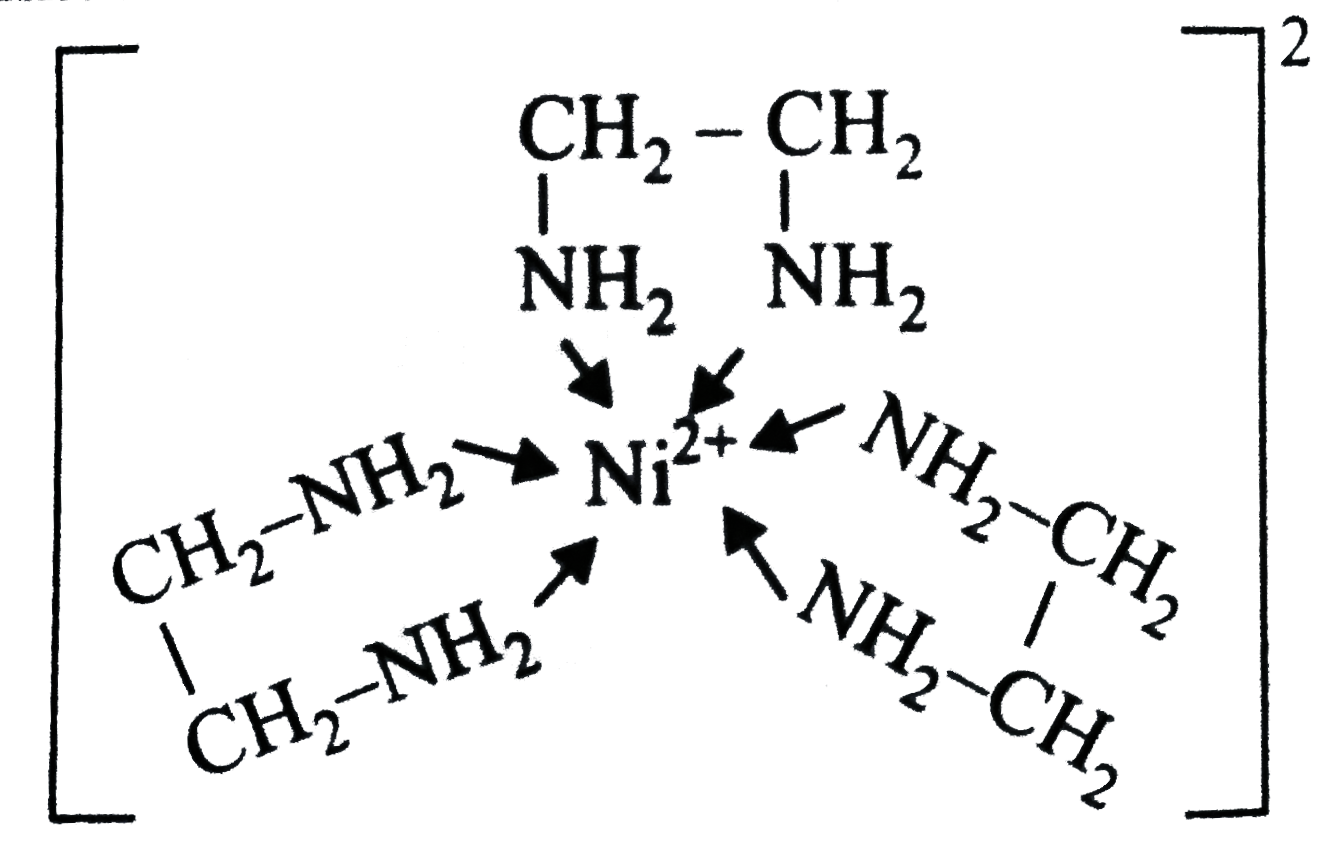
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
COORDINATION COMPOUNDS
A2Z|Exercise AIPMT/NEET Questions|57 VideosCOORDINATION COMPOUNDS
A2Z|Exercise AIIMS Questions|17 VideosCOORDINATION COMPOUNDS
A2Z|Exercise Metal Carbonyls, Stability Of Complexes And Applications|23 VideosCHEMICAL KINETICS
A2Z|Exercise Section D - Chapter End Test|30 VideosELECTROCHEMISTRY
A2Z|Exercise Section D - Chapter End Test|30 Videos
Similar Questions
Explore conceptually related problems
A2Z-COORDINATION COMPOUNDS-Section B - Assertion Reasoning
- The question consist of two statements each, printed as Assertion and ...
Text Solution
|
- The question consist of two statements each, printed as Assertion and ...
Text Solution
|
- The question consist of two statements each, printed as Assertion and ...
Text Solution
|
- These questions consist of two statements each, printed as Assertion a...
Text Solution
|
- The question consist of two statements each, printed as Assertion and ...
Text Solution
|
- The question consist of two statements each, printed as Assertion and ...
Text Solution
|
- The question consist of two statements each, printed as Assertion and ...
Text Solution
|
- The question consist of two statements each, printed as Assertion and ...
Text Solution
|
- The question consist of two statements each, printed as Assertion and ...
Text Solution
|
- The question consist of two statements each, printed as Assertion and ...
Text Solution
|
- The question consist of two statements each, printed as Assertion and ...
Text Solution
|
- The question consist of two statements each, printed as Assertion and ...
Text Solution
|
- The question consist of two statements each, printed as Assertion and ...
Text Solution
|
- The question consist of two statements each, printed as Assertion and ...
Text Solution
|
- The question consist of two statements each, printed as Assertion and ...
Text Solution
|