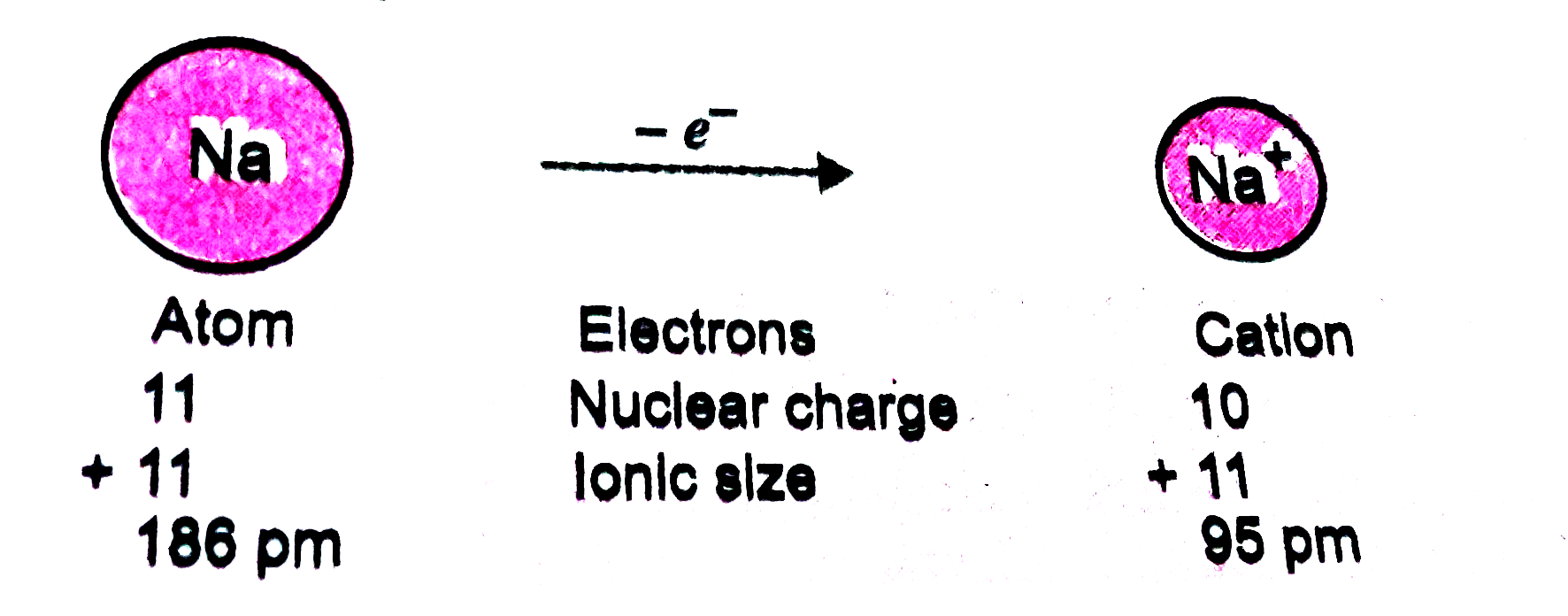Text Solution
Verified by Experts
Topper's Solved these Questions
CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES
PRADEEP|Exercise NCERT (EXEMPLAR PROBLEMS) (Multiple Choice Questions -I)|13 VideosCLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES
PRADEEP|Exercise NCERT (EXEMPLAR PROBLEMS) (Multiple Choice Questions -II)|10 VideosCLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES
PRADEEP|Exercise NCERT (Questions and exercise )(NCERT INTEXT SOLVED QUESTIONS)|10 VideosCHEMICAL BONDING AND MOLECULAR STRUCTURE
PRADEEP|Exercise Curiosity Questions|2 VideosENVIRONMENTAL CHEMISTRY
PRADEEP|Exercise COMPETITION FOCUS (JEE(Main and Advanced)/Medical Entrance (VI.ASSERTION-REASON) Type II|6 Videos
Similar Questions
Explore conceptually related problems
PRADEEP-CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES -NCERT (Questions and exercise )( NCERT EXERCISES)
- Would you expect the second electron gain enthalpy of O as positive, m...
Text Solution
|
- What is the basic difference between the terms electron gain enthalpy ...
Text Solution
|
- How would you react to the statement that the electronegativity of N o...
Text Solution
|
- Describe the theory associated with the radius of an atom as it a. g...
Text Solution
|
- Would you expect the first ionisation enthalpies for two isotopes of t...
Text Solution
|
- What are the major differences between metals and non-metals?
Text Solution
|
- Use the periodic table to answer the following questions. a. Identif...
Text Solution
|
- The increasing order of reactivity among group 1 elements is LiltNaltK...
Text Solution
|
- Write the general outer electronic configuration of s-,p-,d- and f-blo...
Text Solution
|
- Assign the position of the element having outer electronic configurati...
Text Solution
|
- The first (Delta(i)H(1)) and second (Delta(i)H(2)) ionisation enthalpi...
Text Solution
|
- Predict the formula of the stable binary compounds that would be forme...
Text Solution
|
- In the modern periodic table, the period indicates the value of a. a...
Text Solution
|
- Which of the following statements related to the modern periodic table...
Text Solution
|
- Anything that influences the valence electrons will affect the ch...
Text Solution
|
- The size of isoelectronic species -F^(-) ,Ne and Na^(+) is affe...
Text Solution
|
- Which one of the following statements is incorrect in relation to ioni...
Text Solution
|
- Considering the elements B ,A1, Mg and K, the correct order of thei...
Text Solution
|
- Consider a reaction A(g)overset(k=0.1 M min^(-1))to2B(g). If initial c...
Text Solution
|
- A B C D is a parallelogram and E\ a n d\ F are the centroids of tri...
Text Solution
|