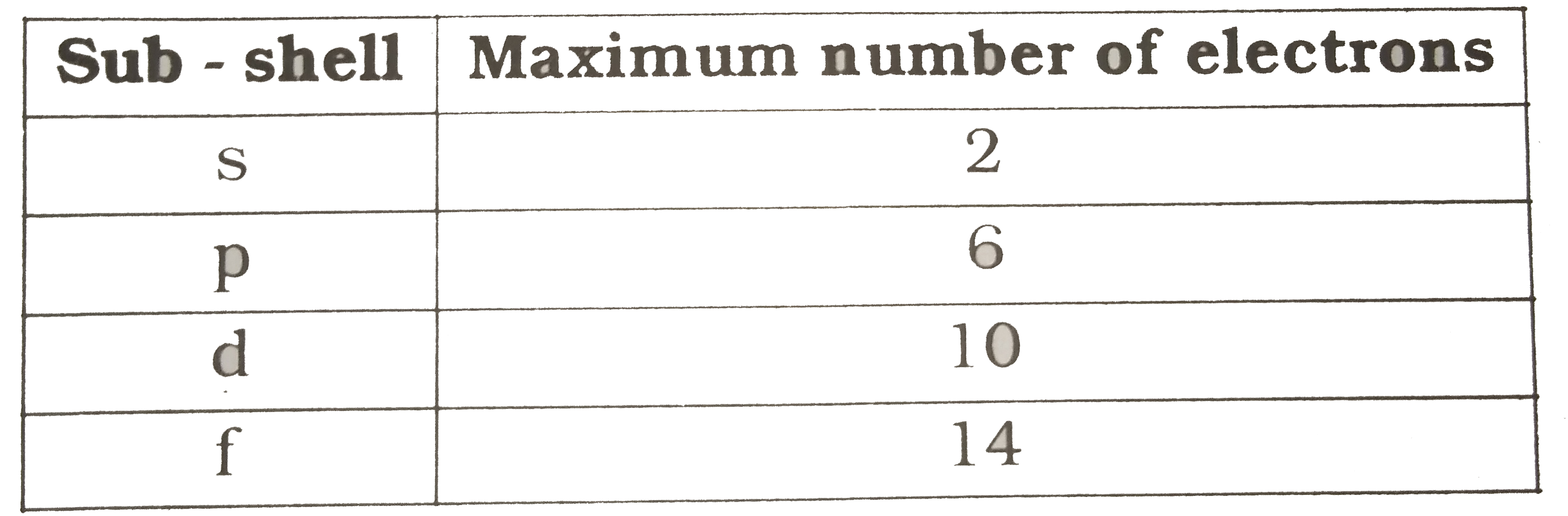Text Solution
Verified by Experts
Topper's Solved these Questions
STRUCTURE OF ATOM
VGS PUBLICATION-BRILLIANT|Exercise CREATIVE QUESTIONS FOR NEW MODEL EXAMINATION ( SECTION - III 2 MARKS QUESTIONS )|38 VideosSTRUCTURE OF ATOM
VGS PUBLICATION-BRILLIANT|Exercise CREATIVE QUESTIONS FOR NEW MODEL EXAMINATION ( SECTION - IV 4 MARKS )|32 VideosSTRUCTURE OF ATOM
VGS PUBLICATION-BRILLIANT|Exercise CREATIVE QUESTIONS FOR NEW MODEL EXAMINATION ( SECTION - I 1/2 MARK QUESTIONS)|96 VideosREFLECTION AND REFRACTION
VGS PUBLICATION-BRILLIANT|Exercise EXERCISE|24 VideosSUMMATIVE ASSESSMENT
VGS PUBLICATION-BRILLIANT|Exercise SUMMATIVE ASSESSMENT|28 Videos
Similar Questions
Explore conceptually related problems
VGS PUBLICATION-BRILLIANT-STRUCTURE OF ATOM -CREATIVE QUESTIONS FOR NEW MODEL EXAMINATION ( SECTION - II 1 MARK)
- Which colours do you observe when an iron rod is gradually heated ?
Text Solution
|
- Manoj observed a street light which is producing yellow light. Then he...
Text Solution
|
- What happens, when an electron. is in excited state ?
Text Solution
|
- Write two sentences about the electron in the 3p^(1) orbital.
Text Solution
|
- How many orbitals there in the orbit n = 3 ? What are they ?
Text Solution
|
- Fill the sub-shells in the given table.
Text Solution
|
- l = 1 is given . What is the shape of the orbital ? Why ?
Text Solution
|
- Name the m 1 values for l = 1.
Text Solution
|
- How many orbitals are there in p-shell ?
Text Solution
|
- What is the relation between sub-shells and no. of electrons in it ?
Text Solution
|
- What information can you write from the given data ?
Text Solution
|
- Draw the Bohr- Sommerfeld model for n= 2
Text Solution
|
- What is the shape of s-orbital ?
Text Solution
|
- Draw p(x) - orbital.
Text Solution
|
- Draw the shape of d(z^(2)) orbital .
Text Solution
|
- What is Sommerfeld's contribution for the structure of atom ?
Text Solution
|
- How do you appreciate the quantum numbers ?
Text Solution
|
- What is the use of a line spectra?
Text Solution
|
- What is the use of atomic spectra ?
Text Solution
|
- Explain the principle which discribes the arrangements of electrons in...
Text Solution
|