A
B
C
D
Text Solution
Verified by Experts
Topper's Solved these Questions
ATOMIC PHYSICS
PHYSICS GALAXY - ASHISH ARORA|Exercise Advance MCQs with One or More Options Correct|30 VideosATOMIC PHYSICS
PHYSICS GALAXY - ASHISH ARORA|Exercise Unsolved Numerical Problems|39 VideosATOMIC PHYSICS
PHYSICS GALAXY - ASHISH ARORA|Exercise Conceptual MCQs Single Option Correct|29 VideosCAPACITANCE
PHYSICS GALAXY - ASHISH ARORA|Exercise UNSOLVED NUMERICAL PROBLEMS|40 Videos
Similar Questions
Explore conceptually related problems
PHYSICS GALAXY - ASHISH ARORA-ATOMIC PHYSICS-NumericalMCQsSingle OptiorisCorrect
- Imagine a neutral particle of same mass m as electron revolving around...
Text Solution
|
- Determine the ratio of perimeters in 2^(nd) and 3^(rd) Bohr orbit inHe...
Text Solution
|
- the photon radiated from hydrogen corresponding to the second line of ...
Text Solution
|
- An alpha-particle with a kinetic energy of 2.1eV makes a head on colli...
Text Solution
|
- A hydrogen atom is initially atrest and free to moveis in the second e...
Text Solution
|
- An gas of H-atom in excited state n(2) absorbs a photon of some energy...
Text Solution
|
- Imagine an atom made of a proton and a hypothetical particle of double...
Text Solution
|
- One of the lines in the emission spectrum of Li^(2+) has the same wave...
Text Solution
|
- In Bohr's theory the potential energy of an electron at a position is ...
Text Solution
|
- If first and second frequencies in transition to orbital are related b...
Text Solution
|
- The ratio of de-Broglie wave length of a photon and an electron of mas...
Text Solution
|
- A monochromatic radiation of wavelength lambda is incident on a sample...
Text Solution
|
- Of the following transitions in a hydrogen atom, the one which gives a...
Text Solution
|
- An electron of the kinetic energy 10 eV collides with a hydrogen atom ...
Text Solution
|
- If the first orbit of a hydrogen atom the total energy of the electron...
Text Solution
|
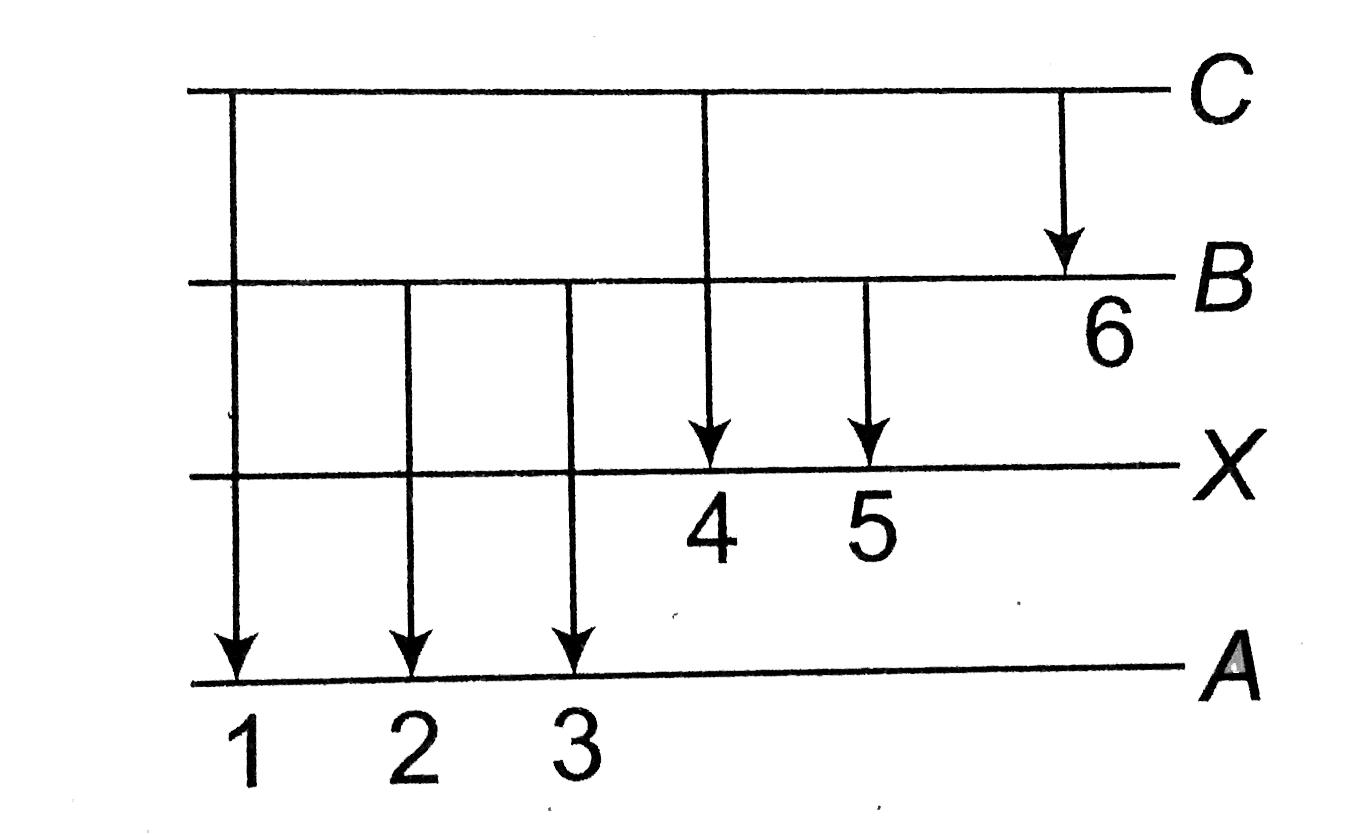
- In the figure six lines of emission spectrum are shown. Which of them ...
Text Solution
|
- An orbit electron in the ground state of hydrogen has an angular momen...
Text Solution
|
- The ratio of the maximum wavlength of the Lyman series in hydrogen spe...
Text Solution
|
- Consider atoms H, He^(+), Li^(++) in their ground states. Suppose E(1)...
Text Solution
|
- The radius of first Bohr orbit of hydrogen atom is 0.53Å. Then the rad...
Text Solution
|